Mastering Lewis Structures: Unveil Chemistry Secrets Today
Are you struggling to understand the fundamentals of chemistry, specifically when it comes to drawing and interpreting Lewis structures? You’re not alone. Many students find Lewis structures to be a challenging concept to grasp, often because they seem abstract and somewhat disconnected from the real-world applications they enable. This guide aims to demystify the process, making it approachable and practical. By breaking down the steps into manageable sections and providing real-world examples, you’ll gain a comprehensive understanding that will serve you well in your chemistry journey.
Why Mastering Lewis Structures Matters
Understanding Lewis structures is crucial for grasping how atoms interact with each other to form molecules. These diagrams, named after the American chemist Gilbert N. Lewis, who introduced the concept in 1916, offer a simple yet powerful way to visualize the arrangement of electrons around atoms. Mastery of this skill opens the door to comprehending chemical bonding, molecular geometry, and the behavior of molecules in various reactions. Whether you're aiming to excel in academic chemistry or simply wish to deepen your understanding of the scientific world, mastering Lewis structures is an essential step.When you face difficulty in grasping complex chemical concepts, it often stems from not fully understanding the foundational aspects. Lewis structures are that foundational aspect for chemistry, providing a clear visual representation of how atoms share electrons to form bonds. This understanding is not just theoretical but has profound implications in real-world applications, such as determining how different compounds react with each other, how they can be synthesized, and even how they affect biological systems.
Quick Reference
Quick Reference
- Immediate action item: Begin with a simple molecule, like water (H2O), to get familiar with the process of drawing Lewis structures.
- Essential tip: Remember the octet rule, which guides the majority of covalent bonding patterns, especially for second-period elements.
- Common mistake to avoid: Forgetting to distribute electrons correctly can lead to an incorrect structure. Always ensure that each atom achieves a stable electron configuration.
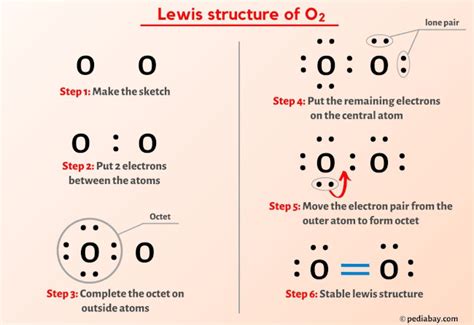
Step-by-Step Guide to Drawing Lewis Structures
Drawing a Lewis structure involves several steps, each critical for ensuring accuracy and clarity in representing molecular structures. Here’s a detailed walkthrough:
Step 1: Count the Total Number of Valence Electrons
The first step in drawing a Lewis structure is to determine the total number of valence electrons in the molecule or ion. This is done by summing the valence electrons of each atom involved in the molecule. For example, in the case of carbon dioxide (CO2), carbon has 4 valence electrons, and each oxygen atom has 6. Thus, the total is 4 (from carbon) + 6 (from each oxygen) = 16 electrons.
Step 2: Determine the Lewis Structure’s Skeleton
The skeleton of a Lewis structure is determined by the placement of the least electronegative atom in the center. This atom will be bonded to the more electronegative atoms surrounding it. In CO2, carbon is less electronegative than oxygen, so it is placed in the center, with the two oxygen atoms surrounding it.
Step 3: Connect Atoms with Single Bonds
Start by placing single bonds between the central atom and each surrounding atom. In CO2, carbon will form a double bond with each oxygen atom to begin the structure.
Step 4: Distribute Remaining Electrons
After forming the bonds, distribute the remaining valence electrons to satisfy the octet rule for each atom, starting with the outer atoms. In CO2, each oxygen atom will first satisfy the octet rule by using the electrons from the double bonds. If any electrons remain, they will be placed on the central atom. In CO2, carbon does not need any additional electrons after bonding, but if more complex structures are considered, this step becomes crucial.
Step 5: Check for Resonance Structures
Some molecules can have more than one valid Lewis structure, known as resonance structures. These structures are different arrangements of electrons that may be represented by a single molecule. For CO2, the resonance structure is simply the same, but in more complex molecules, like nitrate (NO3-), there are different ways to distribute the double bond.
Advanced Lewis Structure Techniques
Once comfortable with basic structures, delve into more complex molecules and ions. These involve understanding exceptions to the octet rule, such as in the case of phosphorus in PCl5, which has more than an octet, or sulfur in SF6, which has an expanded octet.
Handling Molecules with Expanded Octets
In molecules like sulfur hexafluoride (SF6), sulfur has an expanded octet, meaning it can hold more than eight electrons. This occurs when the central atom is from the third period or beyond, allowing it to have empty d-orbitals that can accommodate additional electrons.
Understanding Molecules with Odd Electrons
Sometimes, a molecule may have an odd number of valence electrons, leading to the presence of a free radical or a molecule with an unpaired electron. Nitrogen dioxide (NO2) is an example where the unpaired electron is part of its Lewis structure, playing a role in its reactivity and color.
Exploring Hypervalent Molecules
Hypervalent molecules, like the ones formed by elements in the third period and beyond, can exceed the traditional octet rule. Phosphorus pentachloride (PCl5) is a classic example where phosphorus has five bonding pairs of electrons, using its available d-orbitals to accommodate more electrons.
Practical FAQ
How can I use Lewis structures to predict molecular shape?
Lewis structures are instrumental in predicting the shape of molecules through their connection to the VSEPR (Valence Shell Electron Pair Repulsion) theory. After drawing the Lewis structure for a molecule, identify the central atom and count its bonding and non-bonding electron pairs. These pairs will repel each other, influencing the molecular geometry. For example, in water (H2O), the two lone pairs on the oxygen atom and two bonding pairs with hydrogen create a bent shape.
What are common errors to avoid when drawing Lewis structures?
A frequent mistake is the incorrect placement of the central atom or not correctly distributing electrons to satisfy the octet rule for all atoms. Always ensure that the least electronegative atom is in the center and that each atom, especially the outer ones, achieves a stable electron configuration, usually an octet or, in the case of hydrogen, a duet (two electrons).
Mastering Lewis structures will not only enhance your understanding of chemical bonding and molecular interactions but also equip you with a tool to predict the properties and behaviors of molecules in various contexts, from laboratory reactions to environmental processes. By approaching the subject methodically and practicing with diverse molecules, you’ll develop the skills needed to tackle more complex chemical challenges confidently.