Understanding the 0th Law of Thermodynamics: A Practical Guide
The 0th Law of Thermodynamics often raises more questions than it answers. Unlike the well-known first, second, and third laws, the 0th law provides a fundamental basis for temperature measurement and thermal equilibrium. This guide will delve into the practical applications of the 0th Law, offering step-by-step guidance to ensure you fully grasp its significance and utility.
Why Understanding the 0th Law Matters
Before we dive into the specifics, let’s address the “why” behind this seemingly obscure concept. The 0th Law of Thermodynamics is essential for anyone working with thermal systems or engaged in scientific research. It underpins the concept of temperature and how it helps us predict and control the behavior of energy within a system. Knowing this law ensures that your experiments, designs, or everyday tasks involving heat are based on a sound foundation.
For example, if you’re an engineer designing a new cooling system, understanding the 0th law will allow you to create a setup that accurately measures and maintains temperature, ensuring efficiency and functionality.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Always calibrate your thermometers before use to ensure accurate readings.
- Essential tip with step-by-step guidance: To calibrate a thermometer, use a known temperature source like ice water (0°C) and boiling water (100°C) to adjust the scale.
- Common mistake to avoid with solution: Assuming that all thermometers provide accurate readings without calibration; this can lead to significant errors in temperature measurement.
What is the 0th Law of Thermodynamics?
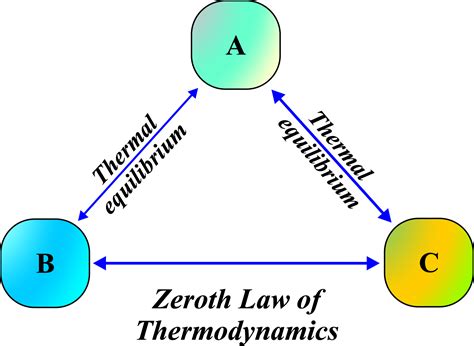
The 0th Law of Thermodynamics states that if three systems are such that each pair is in thermal equilibrium with each other, then they are all in thermal equilibrium with one another. In simpler terms, if System A is in thermal equilibrium with System B, and System B is in thermal equilibrium with System C, then System A must also be in thermal equilibrium with System C. This is foundational for defining temperature and ensuring that a thermometer’s reading is reliable.
Detailed Explanation of the 0th Law
To fully appreciate the 0th Law’s importance, let’s break it down step-by-step:
1. Understanding Thermal Equilibrium: When two systems are in thermal equilibrium, it means they have the same temperature. The exchange of heat between them ceases because there is no net flow of thermal energy.
2. The Role of Thermometers: Thermometers rely on this law to provide accurate temperature readings. When a thermometer is placed in contact with a substance, it reaches thermal equilibrium with that substance, reflecting its temperature.
3. Implications for Science and Engineering: This law’s implications extend to various scientific and engineering fields. For instance, in constructing or using any thermal device, understanding this law ensures that temperature measurements are consistent and reliable.
Consider the example of designing a climate control system for a building. If different sensors (thermometers) are used, the 0th Law ensures that all sensors will agree on the temperature, allowing the system to adjust correctly and maintain the desired environment.
Step-by-Step Application of the 0th Law
To apply the 0th Law of Thermodynamics in real-world scenarios, follow these steps:
Step 1: Identify the Need for Thermal Equilibrium
Determine if your system requires reliable temperature readings. This is crucial in any process where accurate temperature control is essential, such as in chemical reactions, climate control systems, or even everyday cooking.
Step 2: Calibrate Your Thermometer
Before taking any temperature reading, ensure your thermometer is calibrated. Here’s a simple method:
- Using Standard Temperature Points: Use a known temperature reference point, such as ice water (0°C) and boiling water (100°C at sea level) to calibrate your thermometer.
- Adjustment: If your thermometer reads differently at these points, adjust it to reflect the true temperature.
- Repeat: Always calibrate before use, especially if the thermometer has been exposed to different temperature conditions.
Step 3: Ensure Consistent Readings Across Systems
Ensure that all thermometers (or thermal sensors) used in your system are consistent. This requires checking that each system is in thermal equilibrium with another.
For example, in an industrial setup where multiple temperature sensors are used, you might periodically check that all sensors are reading the same temperature using a reference thermometer.
Step-by-step application of the 0th Law ensures that your systems rely on accurate and consistent temperature measurements, leading to efficient and effective operation.
Practical FAQ
Why is calibration important for thermometers?
Calibration is crucial because it ensures that the thermometer provides accurate readings. Without calibration, a thermometer can give incorrect temperature readings, leading to problems in both scientific experiments and everyday applications. For example, an incorrectly calibrated thermometer in a kitchen might give a reading that’s off by a few degrees, affecting cooking times and outcomes. Calibration ensures that the thermometer accurately reflects the temperature of the environment it’s measuring.
How often should I calibrate my thermometer?
The frequency of calibration depends on usage and the importance of accuracy. For routine checks in a kitchen, monthly calibration is usually sufficient. In scientific and industrial applications, it is often recommended to calibrate before each use or at least weekly. High-precision instruments may need daily calibration to maintain accuracy.
What are common mistakes people make with the 0th Law?
One common mistake is assuming that all thermometers are automatically accurate without calibration. Another oversight is neglecting to verify thermal equilibrium among multiple thermometers or sensors. These mistakes can lead to incorrect temperature readings and ineffective system operation. For example, in an industrial setup, using uncalibrated sensors might result in improper production conditions, leading to wasted resources and potential safety hazards.
By understanding and applying the 0th Law of Thermodynamics, you can ensure that your thermal measurements are accurate and reliable, helping to optimize performance in various applications, from everyday tasks to advanced engineering projects.