Welcome to the comprehensive guide on understanding absolute age in geological and archaeological contexts. This guide is designed to unravel the complexities of absolute age determination, making it accessible and easy to grasp. Whether you’re a student, a professional, or just someone curious about the age of Earth’s layers or ancient artifacts, this guide will equip you with the knowledge and tools to dive into the depths of absolute dating methods.
Problem-Solution Opening Addressing User Needs
Understanding the age of geological formations and archaeological finds has always been a fascinating yet challenging aspect of scientific inquiry. Traditionally, determining absolute age has involved navigating complex methodologies and jargon-laden explanations. This can often leave both students and professionals feeling overwhelmed, struggling to see the practical application of these methods in real-world contexts. This guide aims to bridge that gap by providing a clear, step-by-step approach to understanding absolute age determination. With actionable advice, real-world examples, and a focus on solving common user pain points, this guide is your key to unlocking the secrets hidden in the layers of time.
Quick Reference
Quick Reference
- Immediate action item: Start by familiarizing yourself with basic geological and archaeological terms related to absolute age.
- Essential tip: Learn the principle of radiometric dating and how it uses the decay of radioactive isotopes to determine age.
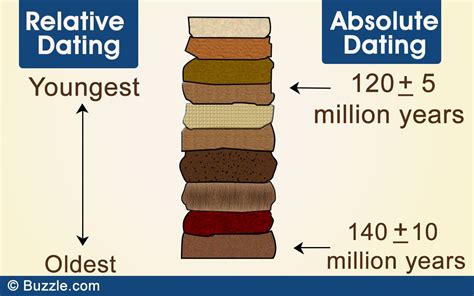
- Common mistake to avoid: Don't confuse absolute age with relative age; absolute age gives a precise date, while relative age shows the chronological order.
Detailed How-To Sections
Understanding Radiometric Dating: A Foundation for Absolute Age Determination
Radiometric dating is the cornerstone of absolute age determination. It involves measuring the decay rate of radioactive isotopes within a sample to estimate how long the sample has been stable since it was formed. Here’s a detailed step-by-step guide on how this process works:
Step 1: Identify the Isotope
To start with radiometric dating, you first need to identify which radioactive isotope you will use. Different isotopes are suitable for dating different types of materials and time ranges. For example, Carbon-14 is ideal for dating organic materials up to about 50,000 years old, while Uranium-Lead dating is best for rocks and minerals that are millions to billions of years old.
Step 2: Sample Collection
The next step involves collecting a sample from the object or layer of interest. For geological samples, this might mean extracting a rock core. For archaeological finds, this could involve carefully excavating and isolating an artifact or fossil.
Step 3: Measurement of Isotope Decay
Using specialized equipment like mass spectrometers, scientists measure the ratio of the parent isotope to the daughter isotope in the sample. For instance, in Carbon-14 dating, the ratio of Carbon-14 to stable Carbon-12 is measured to calculate how long it has been since the organism died.
Step 4: Calculate Half-Life
Each radioactive isotope has a known half-life – the time it takes for half of the parent isotope to decay into the daughter isotope. By applying the principles of radioactive decay and using the decay constant, scientists can calculate the age of the sample. For example, in Carbon-14 dating, the decay constant is used in the formula:
Age = (Ln(N₀/N) × T½) / ln(2)
where N₀ is the initial quantity of the isotope, N is the remaining quantity, and T½ is the half-life of the isotope.
Step 5: Interpreting Results
The final step involves interpreting the calculated age. It’s essential to consider factors such as contamination, environmental changes, and accuracy of the equipment used. For instance, Carbon-14 dating results must be calibrated against historical records to account for fluctuations in atmospheric Carbon-14 levels.
By following these steps, you can accurately determine the absolute age of a wide range of samples, from ancient fossils to geological formations.
Optical Stimulation Luminescence (OSL) Dating
Optical Stimulation Luminescence (OSL) dating is another powerful method for determining the absolute age of sediments. OSL dating relies on measuring the amount of luminescence emitted by quartz or feldspar grains when exposed to light after being buried and shielded from natural light. Here’s a detailed guide to OSL dating:
Step 1: Sample Collection
Collecting sediment samples is the first step. This involves carefully extracting samples from strata at various depths. It’s crucial to avoid light exposure during collection to maintain the sediment's natural state.
Step 2: Preparation and Measurement
In the laboratory, the samples are prepared by separating the quartz or feldspar grains from other materials. These grains are then exposed to a controlled light source to release stored energy from radiation exposure. The emitted luminescence is measured using specialized instruments to determine the equivalent dose.
Step 3: Calculation and Calibration
The amount of luminescence is related to the time the sediments were last exposed to natural light. By understanding the dose rate (the rate of radiation absorbed by the grains) and applying it to the measured luminescence, the age of the sediments can be calculated. Calibration against known age standards helps refine the accuracy of these dates.
Step 4: Analysis
The final step involves analyzing the results in the context of the archaeological or geological formation. This includes understanding the depositional history, the environmental conditions during the time of burial, and any potential post-depositional disturbances.
OSL dating is particularly useful for dating young sediments, typically up to around 100,000 years, and provides precise insights into the timing of depositional events.
Common FAQ
What is the difference between absolute and relative dating?
Absolute dating provides a specific age for a sample in years, often through methods like radiometric dating. Relative dating, on the other hand, places events in chronological order without providing specific dates. For example, in relative dating, you might determine that one layer of rock is older than another based on their positions without knowing the exact age of either layer.
How accurate are radiometric dating methods?
Radiometric dating methods are highly accurate, often within a few percent error margin. The accuracy depends on the precision of the equipment used and the calibration of the isotope decay constants. Techniques like Uranium-Lead dating for geological formations can be accurate to within 0.1% for rocks that are billions of years old. Calibration against known standards and consideration of environmental factors further enhance accuracy.
Why is it important to avoid light exposure during sample collection for OSL dating?
Light exposure resets the stored radiation dose in quartz and feldspar grains. If a sample is exposed to light before it is analyzed in the lab, the measured luminescence will not accurately reflect the time since the last exposure to natural radiation. Hence, careful collection methods that avoid light exposure are critical for accurate OSL dating.
With this guide, you now have a solid foundation in the principles and methodologies of absolute age determination. From radiometric dating to OSL dating, understanding these techniques will empower you to explore the depths of geological and archaeological history with confidence and precision.