Understanding the C2H4 Lewis structure is a fundamental aspect of studying organic chemistry and molecular geometry. Ethylene, commonly known as ethylene, is a simple hydrocarbon that comprises two carbon atoms bonded together with four hydrogen atoms. This molecular composition, denoted as C2H4, is crucial for a wide range of industrial applications, including the production of polyethylene plastics, antifreeze, and various organic compounds. To comprehend the chemical behavior of ethylene, mastering its Lewis structure is essential.
The Essence of the C2H4 Lewis Structure
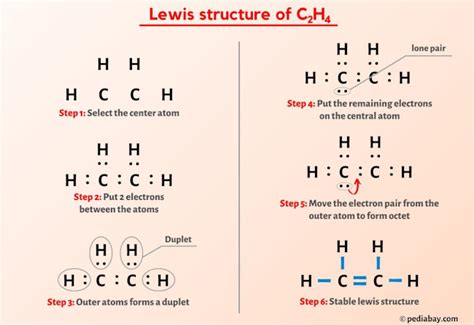
The Lewis structure of ethylene offers a visual representation of the bonding and electron distribution within the molecule. At its core, ethylene is composed of two carbon atoms © that share a double bond. Each carbon atom is also bonded to two hydrogen atoms (H). Drawing this structure involves placing the carbon atoms in the center, with each hydrogen atom attached to a carbon, and the double bond connecting the two carbon atoms.
Steps for Drawing the C2H4 Lewis Structure
- Start by identifying the total number of valence electrons. Carbon has four valence electrons, and each hydrogen has one. Since there are two carbon atoms and four hydrogen atoms, the total number of valence electrons is 12 (from carbons) + 4 (from hydrogens) = 16 electrons.
- Place the two carbon atoms next to each other and connect them with a double bond, using 4 electrons (2 pairs). This accounts for 4 of the 16 valence electrons.
- Allocate the remaining electrons around the carbon atoms. Each carbon atom should have eight electrons, completing their octet. This requires 12 more electrons, which are distributed as single bonds to each hydrogen atom.
Chemical Significance and Real-World Applications
Ethylene’s Lewis structure not only provides insight into its chemical behavior but also highlights its industrial importance. The double bond between the carbon atoms signifies a pi bond in addition to a sigma bond, making ethylene less reactive compared to single-bonded hydrocarbons. This property underpins its stability and versatility in various chemical reactions.
Applications of Ethylene
Ethylene’s unique structure facilitates its role as a key building block in industrial chemistry. It is extensively used in the polymerization process to produce polyethylene, a widely used plastic material in packaging, containers, and piping. Additionally, ethylene is pivotal in producing antifreeze compounds, refrigerants, and numerous organic solvents, demonstrating its versatility and indispensability in modern industry.
Key Insights
- The primary insight with practical relevance is the understanding of ethylene's molecular structure and how it affects its chemical properties and industrial applications.
- A technical consideration with clear application is the double bond in ethylene, which explains its stability and role in polymerization.
- An actionable recommendation is to utilize the Lewis structure of ethylene to predict and control chemical reactions in industrial settings.
Comparative Analysis with Related Molecules
Comparing ethylene with similar molecules like acetylene (C2H2) and formaldehyde (CH2O) provides additional insights. Acetylene, with a triple bond between carbon atoms, is more reactive than ethylene, emphasizing the importance of bond types in molecular stability. Formaldehyde, having a single bond and a carbonyl group, demonstrates the significance of functional groups in chemical reactivity and biological relevance. These comparisons underscore the importance of the C2H4 Lewis structure in understanding broader chemical principles.
FAQ Section
What is the molecular geometry of ethylene?
The molecular geometry of ethylene (C2H4) is planar and trigonal planar around each carbon atom. This results from the double bond between the carbon atoms and the sp2 hybridization.
Why is ethylene important in industrial applications?
Ethylene's stability and reactivity, highlighted by its double bond, make it a crucial precursor in the production of polymers like polyethylene, and other organic compounds used in various industrial applications.
The detailed comprehension of the C2H4 Lewis structure allows for a deeper understanding of ethylene’s role in chemistry and industry. Mastery of this concept enables better prediction and control of chemical reactions, which is vital in both academic research and industrial processes.