Ch2cl2, commonly known as dichloromethane, is an organochlorine compound with widespread applications in chemistry and industrial processes. Its structural simplicity, coupled with a complex molecular geometry, has piqued the interest of many researchers. The question of its polarity is crucial for understanding its solvent properties, reactivity, and interactions in various chemical environments. Here, we dissect the polarity of Ch2cl2, offering expert insights, evidence-based statements, and practical examples to elucidate this complex topic.
Key insights box:
Key Insights
- Ch2cl2 is a polar molecule due to its tetrahedral molecular geometry and electronegativity differences between carbon and chlorine atoms.
- Understanding Ch2cl2’s polarity is critical for predicting its behavior as a solvent in various chemical reactions.
- Useful practical application: Dichloromethane’s polarity makes it effective in dissolving a wide range of organic compounds.
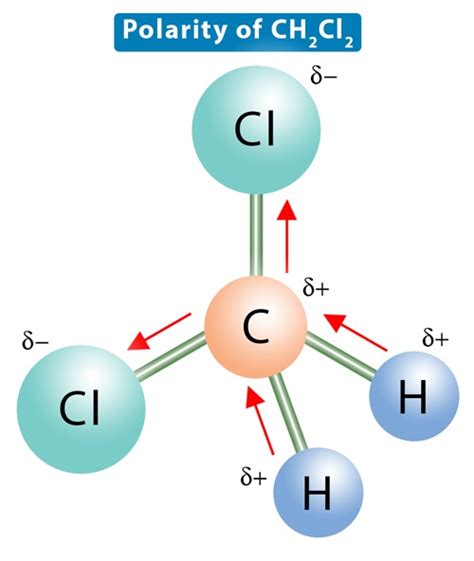
The polarity of Ch2cl2 is inherently tied to its molecular structure and the electronegativity of its constituent atoms. Dichloromethane features a tetrahedral geometry, where a carbon atom is bonded to two hydrogen atoms and two chlorine atoms. The tetrahedral arrangement creates four dipole moments originating from the carbon-halogen (C-Cl) and carbon-hydrogen (C-H) bonds.
In an analysis of the molecular geometry, it becomes evident that the molecule’s symmetrical nature does not negate the individual polarities of the C-Cl and C-H bonds. The electronegativity of chlorine (3.16) is significantly higher than that of carbon (2.55), resulting in a net dipole moment pointing away from the hydrogen atoms and toward the chlorine atoms. Although the C-Cl bonds are symmetrical, their combined effect contributes to an overall polar molecule due to the asymmetric charge distribution. This polarity is a pivotal factor in dichloromethane’s solvent properties, enhancing its ability to dissolve many organic compounds.
In a second analysis, we delve deeper into the implications of dichloromethane’s polarity. The solvent’s capacity to dissolve substances stems from its ability to interact with the solute through dipole-dipole interactions. Given its polar nature, Ch2cl2 can effectively interact with other polar molecules, thus finding utility in various chemical processes, from extractions to reactions requiring polar solvents. For instance, in organic synthesis, dichloromethane often acts as a solvent for reactions that favor the formation of polar products, owing to its own polar characteristics.
A pertinent example illustrating Ch2cl2’s polarity in action is its use in the extraction of chlorophyll from plant materials. The compound’s polarity allows it to interact favorably with the polar functional groups in chlorophyll, thereby facilitating its effective extraction from complex matrices.
FAQ section:
Is dichloromethane’s polarity advantageous in chemical reactions?
Yes, dichloromethane’s polarity makes it an advantageous solvent for a variety of organic reactions, particularly those involving polar compounds. Its ability to dissolve a wide range of substances enhances its utility in both laboratory and industrial applications.
How does the polarity of Ch2cl2 influence its environmental impact?
The polarity of dichloromethane, while beneficial for its solvent properties, also influences its environmental behavior. As a volatile organic compound (VOC), it can contribute to air pollution and has been identified as a substance of environmental concern due to its potential for atmospheric degradation and its impact on human health.
In summary, dichloromethane’s polarity is a fundamental aspect that determines its solvent characteristics and reactivity in chemical processes. Its tetrahedral molecular geometry and significant electronegativity differences contribute to a net dipole moment, making it an effective polar solvent. Understanding these properties provides valuable insights into its practical applications and environmental considerations.