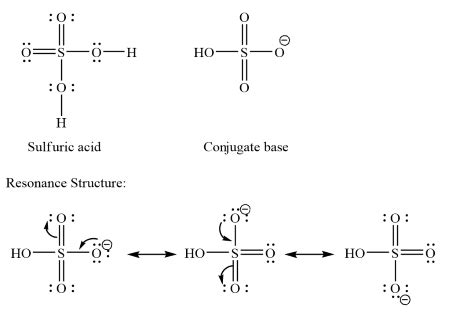
Have you ever wondered what the conjugate base of sulfuric acid (H₂SO₄) is and why it’s significant? This guide is tailored for anyone who is curious about chemical interactions and how acids and bases work together in the world of chemistry. Whether you are a student, a chemistry enthusiast, or just someone who wants to understand a bit more about the molecules that make up our everyday lives, this guide will demystify the conjugate base of H₂SO₄ in a straightforward and actionable manner.
To begin with, let's address a common dilemma many face: understanding the basics of acids, bases, and their conjugates can be overwhelming. However, armed with this guide, you’ll not only grasp the concept but also apply it practically in your studies or daily tasks.
Understanding Acids, Bases, and Conjugates
Acids are substances that donate hydrogen ions (H⁺) when dissolved in water. A classic example is sulfuric acid (H₂SO₄). Bases, on the other hand, are substances that accept hydrogen ions (H⁺). The relationship between an acid and its ability to donate H⁺ is crucial for understanding its conjugate base.
A conjugate base is formed when an acid donates a hydrogen ion (H⁺). For sulfuric acid (H₂SO₄), once it donates a hydrogen ion, it forms the bisulfate ion (HSO₄⁻). So, in simple terms, the conjugate base of H₂SO₄ is HSO₄⁻. This relationship is vital in understanding acid-base reactions, which play a fundamental role in various chemical processes and applications.
Quick Reference
Quick Reference
- Immediate action item: Write down the formula for the conjugate base of H₂SO₄. It is HSO₄⁻.
- Essential tip: Remember, the conjugate base is formed when an acid donates a hydrogen ion. This makes HSO₄⁻ the conjugate base of H₂SO₄.
- Common mistake to avoid: Confusing the conjugate base with the original acid. Always think, “What remains after the acid loses a hydrogen ion?”
How to Determine the Conjugate Base
Determining the conjugate base of an acid is a foundational concept in chemistry. Here’s a step-by-step guide to understanding and applying this process:
To determine the conjugate base of any acid, you should first understand the role of protons (H⁺ ions) in acidic solutions. When an acid is dissolved in water, it donates one or more protons to the water molecules, leading to the formation of its conjugate base.
- Identify the Acid: Start by identifying the acid in question. In our case, it’s H₂SO₄.
- Understand Proton Donation: Understand that acids donate hydrogen ions (H⁺). For H₂SO₄, the first proton donation forms HSO₄⁻, and the second donation forms SO₄²⁻.
- Write Down the Conjugate Base: The conjugate base is formed when the acid donates a proton. For H₂SO₄, after donating one hydrogen ion (H⁺), it forms HSO₄⁻.
- Verify Your Result: Confirm the conjugate base by checking whether the remaining species accepts a proton to revert back to the original acid. Here, HSO₄⁻ can donate another proton to form H₂SO₄.
Through this detailed method, you can systematically determine the conjugate base of any given acid.
The Role of Sulfuric Acid in Reactions
Sulfuric acid (H₂SO₄) is a strong acid and an important player in various chemical reactions. In industry and laboratories, it is used extensively due to its ability to donate two hydrogen ions in a stepwise manner.
In the first step, H₂SO₄ donates one hydrogen ion to water, forming the bisulfate ion (HSO₄⁻) and hydronium ion (H₃O⁺). This reaction is represented as:
H₂SO₄(aq) + H₂O(l) ⇌ HSO₄⁻(aq) + H₃O⁺(aq)
In the second step, HSO₄⁻ can donate another hydrogen ion to form the sulfate ion (SO₄²⁻) and hydronium ion (H₃O⁺). This is depicted as:
HSO₄⁻(aq) + H₂O(l) ⇌ SO₄²⁻(aq) + H₃O⁺(aq)
Understanding these reactions is crucial in fields like analytical chemistry, where precise pH control is necessary.
Why Knowing the Conjugate Base is Important
Knowing the conjugate base of an acid like H₂SO₄ is not just an academic exercise. It has practical implications in understanding acid-base reactions, buffer solutions, and even in industrial processes. Here’s why it matters:
- Buffer Solutions: Buffers maintain pH stability by neutralizing added acids or bases. Knowing the conjugate base helps in creating effective buffer solutions.
- Industrial Chemistry: In processes like pickling, manufacturing of fertilizers, and petroleum refining, understanding conjugate bases is crucial for effective chemical management.
- Environmental Chemistry: Acid-base reactions play a role in environmental chemistry, like the neutralization of industrial waste. Knowledge of conjugate bases aids in managing these processes efficiently.
Practical FAQ
What happens when H₂SO₄ fully ionizes in water?
When sulfuric acid fully ionizes in water, it donates two hydrogen ions in successive steps. Initially, it forms the bisulfate ion (HSO₄⁻) and hydronium ion (H₃O⁺). In the second step, HSO₄⁻ further donates a proton to form the sulfate ion (SO₄²⁻) and another hydronium ion (H₃O⁺). The fully ionized form is H⁺ and SO₄²⁻ in solution.
How does the conjugate base affect the strength of an acid?
The strength of an acid is inversely related to the strength of its conjugate base. A strong acid has a weak conjugate base. Since HSO₄⁻ is the conjugate base of H₂SO₄, and H₂SO₄ is a strong acid, HSO₄⁻ is relatively weak in accepting protons. This explains why H₂SO₄ fully ionizes in the first step, leaving HSO₄⁻ to play a minor role in further ionization.
This guide has delved into the concept of the conjugate base of sulfuric acid, illustrated with step-by-step procedures and practical applications. By understanding these fundamental aspects of acids and bases, you can approach more complex chemical challenges with confidence and precision.
Remember, the journey through chemistry starts with understanding basic concepts, like identifying conjugate bases, which can unlock a myriad of advanced applications and innovations.