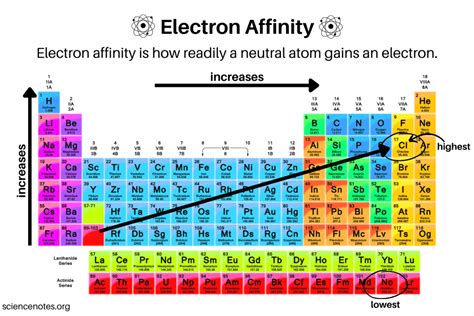
Understanding electron affinity trends is essential for chemistry enthusiasts as it provides a deep insight into how elements interact and form compounds. This exploration reveals not just the behavior of elements but also their reactivity and stability when they gain electrons. Electron affinity, the energy change when an electron is added to a neutral atom in its gaseous state, is a fundamental concept that dictates the nature of chemical bonds and reactions. This article aims to break down these trends and provide expert insights to bolster your chemistry knowledge.
Primary Insights
Key Insights
- Primary insight with practical relevance: The trend of electron affinity generally decreases down a group and increases across a period in the periodic table.
- Technical consideration with clear application: Halogens exhibit high electron affinity due to their tendency to achieve a stable octet configuration.
- Actionable recommendation: To grasp electron affinity trends effectively, one should compare elements’ electronegativities and ionisation energies.
Why Trends in Electron Affinity Matter
Electron affinity trends reveal how atoms interact with additional electrons. A crucial insight is that electron affinity tends to decrease as we move down a group within the periodic table. This trend is primarily due to the increasing atomic radius; as atoms grow larger, the added electron is further from the nucleus and experiences less attraction, hence lower electron affinity. Conversely, moving across a period, electron affinity generally increases due to the added electron being in a smaller orbital closer to the nucleus and experiencing stronger attraction. This principle helps in predicting how reactive elements will be and understanding chemical bonding preferences.Electron Affinity and Chemical Reactivity
Electron affinity plays a critical role in determining the reactivity of elements. For instance, halogens such as fluorine, chlorine, and bromine have high electron affinity values. This high value arises because these elements are just one electron shy of achieving a stable noble gas configuration. As a result, they are highly reactive and tend to form negative ions by gaining an electron. This characteristic is not only essential for understanding halogen reactivity but also for grasping larger chemical reactions involving these elements. For example, chlorine’s strong electron affinity means it readily forms chloride ions, playing a significant role in various industrial and biological processes.Frequently Asked Questions
Why does electron affinity vary across a period?
Across a period, electron affinity generally increases due to the added electron being in a smaller orbital closer to the nucleus, experiencing stronger nuclear attraction.
What influence does atomic size have on electron affinity?
Larger atoms in a group have lower electron affinity as the added electron is further from the nucleus and experiences less attraction.
This detailed understanding of electron affinity trends arms you with the ability to predict and comprehend the behavior of elements in chemical reactions, thus fostering a deeper appreciation and expertise in chemistry.