Freezing Point Celsius: Unveil Hidden Insights for Accurate Measurements
Understanding the freezing point of water at Celsius is fundamental for many fields, from culinary arts to scientific research and everyday life. Misunderstanding or miscalculations here can lead to major issues, whether it’s ensuring your ice cream is properly frozen or achieving precise scientific results. This guide will help you navigate the complexities surrounding the freezing point Celsius, providing practical, actionable advice to enhance accuracy and solve common problems you may encounter.
This guide starts by addressing a prevalent issue that many users face: inconsistent or inaccurate measurements of the freezing point. Misalignment in this area can lead to either overly soft or frozen products, especially in cooking and scientific experiments. We aim to decode the intricacies of achieving the exact freezing point of water at 0°C, delivering step-by-step guidance, real-world examples, and expert tips to bolster your understanding and improve your results.
Quick Reference
Quick Reference
- Immediate action item: Always calibrate your thermometer before taking any measurements to ensure accuracy.
- Essential tip: Use a controlled environment, such as an ice bath, for measuring the freezing point precisely.
- Common mistake to avoid: Not removing air bubbles from the sample can skew measurements and should be avoided.
Understanding the Freezing Point Celsius
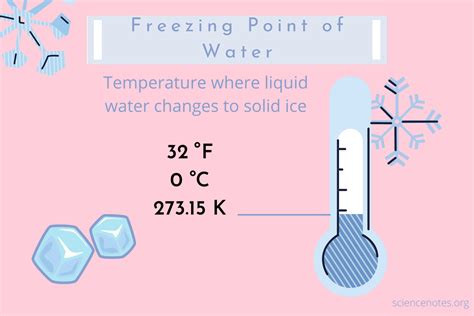
Water’s freezing point at 0°C is a critical benchmark, but what exactly does this mean? When water reaches this temperature, it transitions from a liquid to a solid state under the right conditions. The stability of this transition depends on a variety of factors, including pressure and the presence of impurities. Let’s delve deeper into the specifics to ensure you have a firm grasp on this concept.
Why Accuracy Matters
Accuracy in measuring the freezing point is paramount for several reasons:
- Scientific Precision: Many scientific experiments and calibration processes rely on precise measurements of the freezing point.
- Food Safety and Quality: Accurate freezing temperatures are crucial in preserving food quality and ensuring safety.
- Industrial Applications: Industries such as pharmaceuticals and food production depend on consistent freezing temperatures to ensure product efficacy.
How to Measure the Freezing Point Accurately
Achieving an accurate measurement of the freezing point requires careful preparation, attention to detail, and the right equipment. Let’s walk through the steps to ensure your readings are precise and reliable.
Step-by-Step Guide
Here’s a detailed approach to measuring the freezing point accurately:
1. Gather Your Materials
Before starting, make sure you have the following items on hand:
- A calibrated digital thermometer
- An ice bath (made with ice and water)
- A beaker or container
- Pure water (deionized if possible)
- A stirring rod
- Safety goggles
2. Prepare the Ice Bath
An ice bath is essential for accurately achieving the freezing point. Here’s how to create one:
- Fill a large container (like a large bowl) with ice and add several cups of water.
- Mix the ice and water thoroughly to ensure a homogenous mixture.
- Place this mixture in a stable environment where it won’t spill easily.
3. Calibrate Your Thermometer
To ensure your readings are accurate, calibrate your thermometer:
- Place the thermometer in the ice bath without touching the bottom.
- Wait until the thermometer stabilizes at 0°C.
- If necessary, adjust your thermometer to match this reading.
4. Prepare the Sample
To accurately measure the freezing point of water, use pure, deionized water:
- Fill the beaker with approximately 100 ml of pure water.
- Make sure to remove any air bubbles, as these can interfere with the measurement.
5. Conduct the Measurement
Follow these steps to conduct the measurement accurately:
- Place the beaker in the ice bath.
- Use a stirring rod to gently stir the water in the beaker, ensuring that it cools evenly.
- Monitor the thermometer consistently, looking for the first indication of frost forming on the beaker.
- Record the temperature when the first ice crystal forms. This should be around 0°C.
Best Practices for Accuracy
To enhance the accuracy of your measurements, consider these best practices:
- Ensure your thermometer is fully submerged without touching the bottom of the container.
- Use high-quality, pure water to avoid impurities affecting the freezing point.
- Conduct measurements in a stable environment to prevent temperature fluctuations.
- Regularly calibrate your thermometer to maintain precision.
Common Pitfalls and How to Avoid Them
Even with careful preparation, certain pitfalls can arise that affect accuracy. Here’s how to avoid them:
1. Air Bubbles
Air bubbles can elevate the freezing point and give inaccurate readings.
- Solution: Gently tap the container to release bubbles or use a needle to pop them if necessary.
2. Thermometer Placement
Improper thermometer placement can skew results.
- Solution: Ensure the thermometer is fully submerged and not touching the container’s bottom.
3. Temperature Fluctuations
Sudden temperature changes can cause inaccuracies.
- Solution: Conduct measurements in a controlled, stable environment.
Practical FAQ
How do I know if my thermometer is calibrated?
To determine if your thermometer is calibrated, place it in an ice bath. It should read 0°C when the ice and water mixture is at equilibrium. If it doesn’t match, adjust your thermometer to align with this temperature.
What type of water should I use for freezing point measurement?
For the most accurate results, use pure, deionized water. Regular tap water contains impurities that can affect the freezing point measurement.
Why does my ice bath not reach 0°C?
If your ice bath doesn’t reach 0°C, ensure the ice is fresh and the container is large enough to maintain a stable temperature. Avoid opening the container frequently as it will cause temperature fluctuations.
Advanced Tips for Professionals
For those needing even more precision, here are advanced tips that can further refine your measurement accuracy:
1. Using a Standard Reference Material
In scientific work, using a standard reference material ensures ultra-precise measurements:
- Obtain a certified standard reference material from a reputable lab.
- Compare your measurements to this standard to validate accuracy.
2. Automated Measurement Systems
For laboratories requiring frequent precise measurements:
- Invest in automated systems that constantly monitor and adjust for external temperature changes.
- These systems can provide more reliable and reproducible results over time.
3. Advanced Calibration Techniques
For the most exacting standards: