Understanding enthalpy calculations is crucial for scientists, engineers, and students in the field of chemistry and thermodynamics. Enthalpy, a fundamental concept in thermodynamics, represents the total heat content of a system. Mastering enthalpy calculations can streamline your work and provide invaluable insights into the energy changes within a system. This guide is designed to be your ultimate resource for understanding and performing enthalpy calculations accurately.
From basic principles to advanced techniques, this guide covers everything you need to know. Whether you're trying to grasp the foundational concepts for the first time or aiming to refine your existing knowledge, this comprehensive resource provides step-by-step guidance, practical examples, and actionable advice to tackle any enthalpy-related problem you encounter.
Understanding Enthalpy: The Essentials
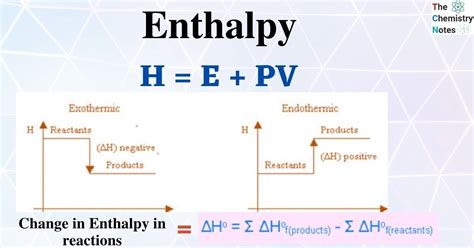
Enthalpy (H) is the measure of the total heat content of a thermodynamic system. It’s especially useful in the context of chemical reactions and phase changes, where heat exchange is a key factor. To calculate enthalpy, you typically consider two main components: the internal energy of the system (U) and the product of pressure (P) and volume (V) that the system occupies.
The formula for enthalpy is:
H = U + PV
This basic equation encapsulates the total heat content within a system, making it a pivotal concept for understanding energy transfer during chemical processes.
Quick Reference: Key Enthalpy Calculation Tips
Quick Reference
- Immediate action item with clear benefit: Start with standard enthalpy change of formation (ΔHf°) values from tables, as these provide a solid base for calculating enthalpy changes in reactions.
- Essential tip with step-by-step guidance: Use Hess's Law for indirect enthalpy calculations. Hess’s Law states that the total enthalpy change for a reaction is the same, regardless of the path taken. This can help simplify complex calculations.
- Common mistake to avoid with solution: Ensure unit consistency. Always convert all values to the same units (e.g., kJ/mol) before performing calculations to avoid errors.
Detailed How-To Sections: Calculating Enthalpy Step-by-Step
Step 1: Understanding Enthalpy Change in Reactions
To begin, it’s important to understand the concept of enthalpy change (ΔH) in a chemical reaction. Enthalpy change is the difference in enthalpy between the products and the reactants of a chemical reaction. It’s usually expressed in kJ/mol. Here’s a simple way to conceptualize it:
ΔH = H(products) - H(reactants)
The enthalpy change indicates whether a reaction is endothermic (absorbing heat, ΔH > 0) or exothermic (releasing heat, ΔH < 0).
Step 2: Using Standard Enthalpy of Formation
For many enthalpy calculations, we use standard enthalpy of formation (ΔHf°). This is the change in enthalpy when one mole of a substance is formed from its elements in their standard states under standard conditions (298.15 K and 1 atm).
To calculate the total enthalpy change of a reaction using standard enthalpy of formation values:
- Write down the balanced chemical equation for the reaction.
- Determine the standard enthalpy of formation for each reactant and product.
- Multiply the standard enthalpy of formation by the number of moles of each substance.
- Calculate the total enthalpy change using the equation: ΔH = Σ [H(products)] - Σ [H(reactants)].
Here's an example to solidify your understanding:
Consider the formation of water from hydrogen and oxygen:
2H₂(g) + O₂(g) → 2H₂O(l)
Using standard enthalpy of formation values:
| Substance | ΔHf° (kJ/mol) |
|---|---|
| H₂(g) | 0 (by definition) |
| O₂(g) | 0 (by definition) |
| H₂O(l) | -285.8 kJ/mol |
Now calculate:
ΔH = [2 × (-285.8)] - [2 × 0 + 0] = -571.6 kJ
This negative value indicates that the formation of water is an exothermic reaction.
Step 3: Applying Hess’s Law for Complex Reactions
Hess’s Law is an indispensable tool for calculating enthalpy changes in reactions that are not straightforward to measure directly. When you can’t measure the enthalpy change of a reaction directly, Hess’s Law allows you to determine it by adding together the enthalpy changes of individual steps that make up the overall reaction.
Here’s how to apply Hess’s Law:
Step 1: Write down the individual steps of the reaction whose enthalpy changes are known.
Step 2: Reverse any steps that need to be reversed to match the desired reaction.
Step 3: Add the enthalpy changes of all the individual steps to find the total enthalpy change for the overall reaction.
Example:
Suppose we want to find the enthalpy change for the reaction:
N₂(g) + 3H₂(g) → 2NH₃(g)
We can break this down using reactions with known ΔH values:
| Reaction Steps | ΔH (kJ) |
|---|---|
| N₂(g) + 3H₂(g) → 2NH₃(g) | Unknown |
| N₂(g) → 2N(g) | 941.4 kJ |
| 3/2H₂(g) → 3H(g) | 1.5 × 435.8 = 653.7 kJ |
| 2N(g) + 3H(g) → 2NH₃(g) | -330.0 kJ |
Using Hess’s Law:
ΔH(total) = 941.4 + 653.7 - 330.0 = 1265.1 kJ for the decomposition of 2 moles of NH₃.
To find the enthalpy change for the formation of 1 mole of NH₃:
ΔH(1 mole NH₃) = 1265.1 kJ / 2 = 632.55 kJ
Practical FAQ: Addressing Common User Questions
What is the best method to find the standard enthalpy of formation for complex compounds?
For complex compounds, the best method is to consult thermodynamic data tables provided by reliable scientific sources such as the NIST Chemistry WebBook. These tables list standard enthalpy of formation values for a vast number of compounds. For more complex organic or biological molecules, experimental measurements or computational chemistry methods (like quantum mechanics) can provide accurate values.
Always ensure that the data you’re using is up-to-date and pertains to standard conditions (298.15 K and 1 atm). When tables don’t provide values directly, consider breaking down the compound into simpler molecules and using Hess’s Law to calculate it indirectly.
How can I ensure the accuracy of my enthalpy calculations?
Accuracy in enthalpy calculations relies on precise measurements and attention to unit consistency. Always double-check that all values you use (like temperatures, pressures, and concentrations) are in compatible units. Use standardized