Finding density accurately is crucial in various fields like chemistry, physics, materials science, and even everyday life. Knowing how to calculate density—defined as mass per unit volume—will empower you to understand the properties of different materials and make informed decisions in numerous situations. This guide will walk you through everything you need to know to master the concept and practical application of density.
Introduction to Density
Density is a fundamental property that describes how much mass is contained within a given volume. Knowing the density of a substance can help you identify materials, compare their properties, and understand physical phenomena. This guide will break down the steps to calculate density, highlight essential tips, and offer solutions to common pitfalls. Whether you’re a student, a professional, or just curious, you’ll find practical advice and real-world examples to help you understand and utilize density effectively.
Why Density Matters
Understanding density is essential because it helps us make sense of the world around us. For example, why does oil float on water? Why do objects of different materials behave differently when dropped from the same height? Density answers these questions by providing a measure of how tightly packed the mass is within a given volume. Accurately determining density can also be crucial in industries like manufacturing, where material selection is key to product performance.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Use an analytical balance for precise mass measurement.
- Essential tip with step-by-step guidance: Accurately measure volume using a graduated cylinder or a pycnometer.
- Common mistake to avoid with solution: Ensure the container used to measure volume is dry and free from residue.
Step-by-Step Guide to Calculating Density
Calculating density involves measuring the mass and volume of an object and then dividing the mass by the volume. Here’s a detailed step-by-step process to ensure accuracy.
Gathering Equipment
To calculate density, you will need:
- An analytical or digital balance for mass measurement.
- A graduated cylinder or a pipette for volume measurement.
- An appropriate container for submerging the object to measure its volume.
- Water or another liquid for displacement method.
Measuring Mass
Start by measuring the mass of the object using an analytical balance. Here’s how to ensure accuracy:
- Ensure the analytical balance is on a flat, stable surface.
- Place a container on the balance and tare it to zero.
- Place the object in the container and record the mass displayed.
Measuring Volume
There are several methods to measure volume accurately:
Method 1: Displacement Method
For irregularly shaped objects, use the displacement method:
- Fill a graduated cylinder with a known volume of water (e.g., 100 mL).
- Carefully submerge the object in the water without spilling.
- Measure the new water level in the cylinder.
- Calculate the volume of the object by subtracting the initial volume from the final volume.
Method 2: Graduated Cylinder
For small or regular-shaped objects, directly measure the volume using a graduated cylinder:
- Dip the object into the graduated cylinder.
- Fill the cylinder with water until it’s full.
- Observe the water level at the calibration mark.
- Subtract the initial water level from the final level to get the volume of the object.
Calculating Density
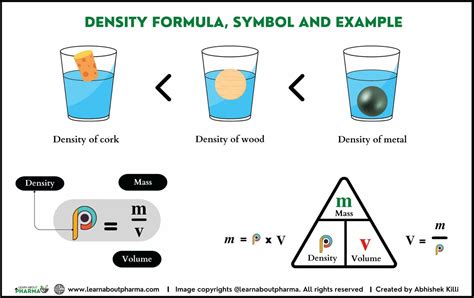
Once you have both the mass and volume, you can calculate the density using the formula:
Density (ρ) = Mass (m) / Volume (V)
For example, if the mass is 50 grams and the volume is 10 mL, the density would be:
ρ = 50 g / 10 mL = 5 g/mL
Best Practices
To ensure accurate density measurements, consider these best practices:
- Calibrate your equipment regularly.
- Avoid air bubbles when measuring volume.
- Use high-quality, clean equipment.
- Double-check your measurements.
Practical Applications of Density
Understanding and calculating density have practical applications across various fields:
- Material Selection: Industries use density to choose materials for specific applications, ensuring that products have the desired properties.
- Food Industry: Chefs and nutritionists use density to determine caloric content and composition of food items.
- Environmental Science: Scientists use density to study ocean currents and the behavior of pollutants in water.
- Medicine: Density is used in medical imaging to identify different tissues within the human body.
Practical FAQ
How do I ensure the accuracy of my density measurements?
Accuracy in density measurements depends on several factors. First, ensure your analytical balance and graduated cylinder are calibrated and in good working condition. Double-check all measurements, and take multiple readings to verify consistency. Avoid air bubbles when measuring volume and ensure that all equipment is clean and dry. Regularly clean and calibrate your measuring instruments to maintain precision.
Can I measure the density of a liquid?
Absolutely, you can measure the density of a liquid using a similar approach. Here’s a simplified method:
- Measure the mass of a container using an analytical balance.
- Fill the container with the liquid to a known volume, marking the level.
- Re-measure the total mass of the container with the liquid.
- Subtract the mass of the container from the total mass to find the liquid’s mass.
- Divide the mass of the liquid by its volume to get the density.
What should I do if my object floats in water?
If your object floats, you can still determine its density, but it requires an alternative approach. Use a hydrometer or a densitometer, which measure the object’s buoyancy in a fluid. Here’s a quick method:
- Use a graduated cylinder filled with water.
- Hang your object from a scale and submerge it in water.
- Record the object’s mass in air and the buoyant force it exerts when submerged.
- Use Archimedes’ principle to calculate the object’s density using the formula:
- Density = Mass / (Volume displaced by the object)
Common Mistakes to Avoid
While calculating density, it’s essential to avoid common pitfalls that can skew your results:
- Air bubbles: Ensure no air is trapped when measuring the volume of an irregular object.
- Improper calibration: Regularly calibrate your equipment to ensure accuracy.
- Inaccurate readings: Double-check your mass and volume measurements.
- Residual contamination: Make sure your measuring container is clean to avoid mixing contaminants with your sample.
Advanced Tips and Techniques
For those looking to delve deeper into density measurement, consider these advanced tips: