Mastering the Lewis Structure for NH3 is essential for understanding the molecular geometry of ammonia, a fundamental molecule in chemistry. This guide offers practical insights and evidence-based statements on how to draw the Lewis structure of NH3, ensuring a clear grasp of its electronic configuration and spatial arrangement.
Understanding the Lewis Structure of NH3
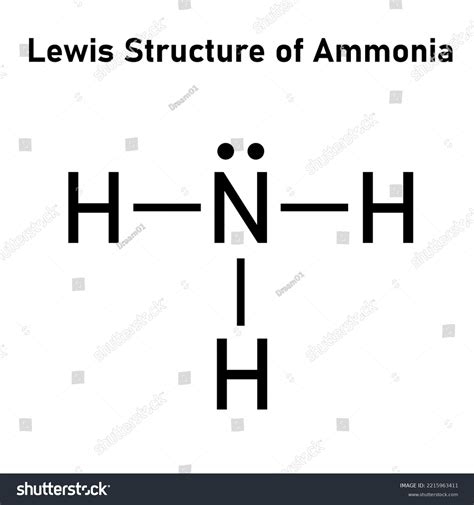
The Lewis structure of NH3 (ammonia) is a crucial aspect of learning about molecular geometries and bonding theories. Ammonia consists of one nitrogen atom bonded to three hydrogen atoms. This simple yet profound molecule is the first step in understanding more complex molecules. By mastering NH3’s Lewis structure, you lay the foundation for exploring more intricate molecular frameworks.
Key Insights
- Primary insight with practical relevance: Ammonia’s trigonal pyramidal molecular geometry is a direct consequence of its Lewis structure.
- Technical consideration with clear application: Knowing NH3's Lewis structure helps in predicting its physical properties and chemical behavior.
- Actionable recommendation: Utilize VSEPR theory alongside the Lewis structure to accurately determine ammonia's molecular geometry.
Drawing the Lewis Structure for NH3
To draw the Lewis structure for NH3, one must first understand the valence electrons. Nitrogen has 5 valence electrons, and each hydrogen has 1. The total is 8 valence electrons. In the NH3 Lewis structure, nitrogen shares one electron with each hydrogen atom, forming three single bonds. This accounts for 6 electrons, leaving 2 electrons to be distributed as a lone pair on the nitrogen atom. This arrangement fulfills the octet rule for nitrogen while allowing each hydrogen to achieve a stable electron configuration.
Understanding Molecular Geometry through NH3’s Lewis Structure
The Lewis structure of NH3 provides a visual representation that informs its molecular geometry. According to the Valence Shell Electron Pair Repulsion (VSEPR) theory, the electron pairs around the central nitrogen atom repel each other, arranging themselves to minimize repulsion. The structure predicts a trigonal pyramidal shape, with the nitrogen at the top and the hydrogen atoms forming the base. The presence of the lone pair on nitrogen contributes to the molecule’s bent shape, influencing its polarity and explaining its relatively high boiling and melting points.
What determines the shape of NH3?
The shape of NH3 is determined by the arrangement of electron pairs around the central nitrogen atom, as predicted by VSEPR theory, along with the presence of a lone pair on the nitrogen atom.
How does the Lewis structure of NH3 affect its chemical properties?
The Lewis structure of NH3 explains its polarity due to the uneven distribution of electron density caused by the lone pair on nitrogen, which makes it a moderately strong base and a weak acid.
This guide underscores the importance of understanding the Lewis structure of NH3 for grasping its molecular geometry and behavior. Through practical insights and evidence-based explanations, you can appreciate how the arrangement of electrons dictates the physical and chemical characteristics of ammonia.