Understanding the molar mass of nitrogen gas (N2) is a fundamental concept in chemistry that applies to numerous real-world contexts, from basic laboratory experiments to industrial processes. This simple guide aims to elucidate the process of determining the molar mass of N2 with practical insights and real examples.
What is Molar Mass?
Molar mass refers to the mass of one mole of a substance. It is a bridge between the atomic scale and the macroscopic world, providing a way to quantify chemical substances. For nitrogen gas, which exists as N2, the molar mass is determined by summing the atomic masses of its constituent atoms.
Calculating the Molar Mass of N2
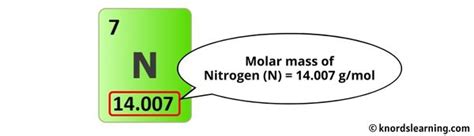
The molar mass of N2 is found by adding the atomic masses of two nitrogen atoms together. According to the periodic table, the atomic mass of a nitrogen (N) atom is approximately 14.01 atomic mass units (amu). Since N2 consists of two nitrogen atoms, the calculation becomes straightforward:
- Atomic mass of N = 14.01 amu
- Molar mass of N2 = 2 × 14.01 amu = 28.02 g/mol
Applications and Practical Relevance
Understanding the molar mass of N2 has several practical applications. For example, in laboratory settings, knowing the molar mass is essential for accurate dilution calculations and for understanding the stoichiometry of chemical reactions. Industrially, nitrogen gas is a critical component in processes such as ammonia production (Haber process), where precise calculations ensure optimal yields and safety.
Key Insights
- Primary insight with practical relevance: Knowing the molar mass of N2 is essential for chemical calculations in both academic and industrial settings.
- Technical consideration with clear application: The molar mass of N2 is used in stoichiometric calculations for processes like ammonia production.
- Actionable recommendation: For science students, mastering the calculation of molar mass will greatly enhance their ability to conduct experiments and understand chemical reactions.
Importance in Real-World Scenarios
Nitrogen gas plays a significant role in various sectors. For instance, in the production of fertilizers, the molar mass of N2 is crucial for ensuring that the correct proportions of nitrogen are used in creating compounds like urea and ammonium nitrate. This precise understanding also helps in determining the quantity of gases required for controlled combustion processes in energy production.
Safety Considerations
In many industrial processes, nitrogen gas is used under high pressure and potentially explosive conditions. Accurate knowledge of the molar mass aids in calculating gas volumes and in ensuring the safety protocols for handling such substances. For instance, in scenarios where nitrogen is used to purge oxygen, knowing the exact molar mass allows for calculating the right amount of N2 gas to ensure complete removal of oxygen, thus preventing hazardous situations.
Why is molar mass important in laboratory experiments?
Molar mass is crucial for accurate preparation of solutions and for understanding the proportions of reactants and products in chemical reactions. It helps ensure precise measurements and successful outcomes in laboratory settings.
In conclusion, understanding the molar mass of N2 is not just a theoretical exercise; it has tangible implications in various scientific and industrial fields. Mastery of this concept is essential for any science student aiming to excel in chemistry and related disciplines.