Understanding Molecular Shape for CH4: Surprising Insights Unveiled
Are you curious about the molecular shape of methane (CH4) and why it’s shaped the way it is? Understanding molecular shape isn’t just a dry academic exercise; it has real-world applications in fields like chemistry, materials science, and even environmental science. Knowing the shape of a molecule like CH4 can help you understand its properties, reactivity, and even how it interacts with other molecules.
Problem-Solution Opening Addressing User Needs
When we talk about molecular shape, we often find ourselves facing a few common roadblocks. For starters, many people struggle to visualize how atoms arrange themselves in space. This visualization isn’t just about drawing; it’s about comprehending the three-dimensional structure and the forces that govern it. Whether you’re a student trying to grasp the basics for an exam, a professional in the field, or just someone curious about the molecules that make up the world around us, you need a clear, straightforward guide that breaks down complex ideas into easy-to-understand steps.
This guide aims to do just that. We’ll demystify the molecular shape of methane, offering a step-by-step approach, real-world examples, and actionable tips to make the abstract concept of molecular geometry tangible and easy to grasp. By the end of this guide, you’ll not only understand why methane has the shape it does but also how this insight can be practically applied in various fields.
Quick Reference
Quick Reference
- Immediate action item: Draw the Lewis structure of CH4 to start visualizing its shape.
- Essential tip: Remember that the shape of CH4 is determined by minimizing electron pair repulsion, leading to a tetrahedral arrangement.
- Common mistake to avoid: Confusing bond angles; the real bond angle in a tetrahedral structure is 109.5 degrees, not 90 degrees.
Why Methane Has a Tetrahedral Shape
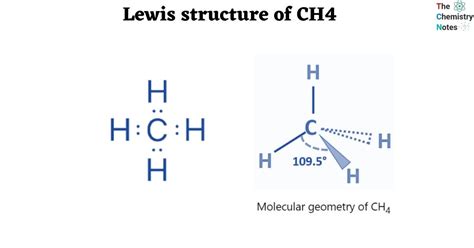
Understanding the shape of methane begins with its Lewis structure. To start, methane consists of one carbon atom bonded to four hydrogen atoms. Here’s how you draw the Lewis structure:
- First, count the total number of valence electrons. Carbon has four, and each hydrogen has one. Therefore, there are 4 (from carbon) + 4 (from four hydrogens) = 8 valence electrons.
- Place the carbon atom in the center and arrange the four hydrogen atoms around it.
- Distribute the electrons in pairs between the carbon and each hydrogen to satisfy the duet rule for hydrogen and the octet rule for carbon. You'll place one electron pair between each hydrogen and the carbon atom.
With all these bonds in place, it’s clear that the carbon atom has four electron pairs to repel each other. The most stable configuration is a tetrahedral shape, where each hydrogen atom occupies a vertex of a tetrahedron, with the carbon at the center. This shape minimizes repulsion between the electron pairs, leading to the molecule’s overall stability.
Detailed How-To Sections
Step-by-Step Guide to Drawing the Lewis Structure for CH4
Drawing the Lewis structure for methane is the first step towards understanding its molecular shape. Let's break it down:
- Count the valence electrons: As previously mentioned, carbon has four valence electrons and each hydrogen has one. So, in total, there are 8 valence electrons for the entire molecule.
- Arrange the atoms: Place the carbon atom in the center and surround it with the four hydrogen atoms.
- Distribute electrons: Start by placing two electrons between the carbon and each hydrogen to form a bond. This accounts for 8 electrons. The remaining electrons will complete the octet around the carbon atom.
- Check octet rule: Ensure that each hydrogen has two electrons (forming a bond), and carbon has eight electrons, fulfilling the octet rule.
Here's a visual representation:
- Carbon (C): ····
- Hydrogen (H): H
Each line represents a single bond:
- C: ···· ····
With all the bonds in place:
- C: :H:
- |
- :H:
By arranging the bonds this way, we fulfill the octet rule for carbon and the duet rule for hydrogen. Now that we have the Lewis structure, let's move on to understanding the tetrahedral shape.
Why the Tetrahedral Shape Minimizes Repulsion
Molecules are not static; they constantly change shape to minimize energy, particularly to minimize repulsion between the electron pairs. In the case of methane, the carbon atom has four electron pairs—two bonding pairs and two lone pairs (though in this case, there are no lone pairs). These pairs repel each other, and the most energy-efficient way for them to do so is to be as far apart as possible. The tetrahedral shape accomplishes this perfectly:
- Each hydrogen atom is positioned at the vertices of a tetrahedron.
- The central carbon atom is at the center of this tetrahedron.
- The bond angles between each hydrogen-carbon-hydrogen pair are approximately 109.5 degrees, providing maximum separation between the electron pairs.
This optimal geometry minimizes repulsive forces, leading to a stable molecular structure.
Advanced Insights into CH4’s Molecular Shape
While the tetrahedral shape is the foundation, diving deeper provides more insights:
- Bond Length and Bond Angle: The bond length in methane is approximately 1.09 Å (angstroms). The bond angles are close to 109.5 degrees, as stated earlier.
- Physical Properties: Methane's tetrahedral shape means it is nonpolar, leading to its characteristic low boiling point and non-reactivity under normal conditions.
- Real-World Implications: Methane's shape and properties make it an excellent non-reactive gas, used in various applications like fuel (natural gas) and even in producing other chemicals.
With these advanced insights, you can appreciate the underlying principles and practical applications of methane’s molecular structure.
Practical FAQ
How does molecular shape affect the properties of CH4?
The molecular shape of CH4 plays a significant role in its properties. The tetrahedral geometry ensures that the molecule is nonpolar, due to the symmetrical distribution of electrons around the central carbon atom. This nonpolar nature results in low boiling and melting points, and minimal reactivity, making methane a stable, non-reactive gas under standard conditions. This stability makes methane ideal for use as a fuel in natural gas pipelines and as a starting point in chemical syntheses.
Why is understanding molecular shape important in chemistry?
Understanding molecular shape is crucial in chemistry because it directly influences the molecule’s physical and chemical properties. The shape determines how a molecule interacts with other molecules, its solubility, reactivity, and even its biological activity if it’s a biochemical compound. For instance, knowing the tetrahedral shape of methane helps predict its low reactivity and non-polar nature, which are critical in applications like fuel and chemical manufacturing.
Can the shape of a molecule like CH4 change under different conditions?
While the basic tetrahedral shape of methane remains constant due to its stable Lewis structure and bond angles, other molecules can undergo shape changes under different conditions, such as changes in temperature or pressure, or due to the presence of different substituents in more complex molecules. For example, a molecule like ammonia (NH3) can change shape slightly based on the electron density around the nitrogen atom.
Conclusion
Understanding the molecular shape of methane (CH4) is more than