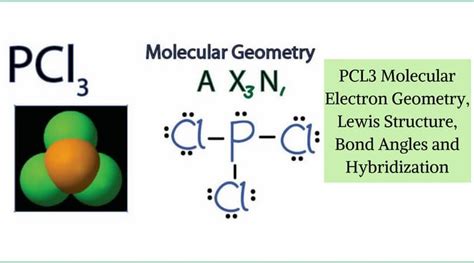
Unveiling the electron geometry of phosphorus trichloride (PCl₃) is vital for understanding molecular structure and reactivity. PCl₃, with a trigonal pyramidal shape, plays a pivotal role in organophosphorus chemistry, impacting fields like agrochemicals and pharmaceuticals. By exploring its electron geometry, we gain deeper insights into its bonding and electronic configuration.
Understanding the Basics of Electron Geometry
Electron geometry considers the spatial arrangement of all electron pairs around a central atom in a molecule. It’s crucial in predicting molecular shapes and interactions. In PCl₃, phosphorus (P) is bonded to three chlorine (Cl) atoms, with an additional lone pair of electrons, defining its electron geometry. The lone pair influences the overall molecular shape, leading to the characteristic trigonal pyramidal form.
Key Insights
- PCl₃ exhibits a trigonal pyramidal electron geometry due to the presence of a lone pair and three bonding pairs around phosphorus.
- The lone pair on phosphorus creates a steric effect that alters the VSEPR geometry.
- Understanding PCl₃'s electron geometry helps predict its reactivity and interactions in chemical reactions.
Molecular Geometry and VSEPR Theory
Valence Shell Electron Pair Repulsion (VSEPR) theory offers a systematic approach to predicting molecular geometry. In PCl₃, phosphorus is the central atom with an expanded octet due to its ability to accommodate more than eight valence electrons. The molecule has four electron pairs around phosphorus: three bonding pairs and one lone pair. VSEPR predicts that these pairs will arrange themselves to minimize repulsion, resulting in a trigonal pyramidal shape with a bond angle of approximately 101.5 degrees.
Electrostatics and Steric Effects
The repulsion between the lone pair and bonding pairs is greater than between bonding pairs alone. This steric effect results in the lone pair occupying more spatial volume, pushing the bonding pairs closer together and flattening the ideal tetrahedral arrangement. Consequently, the bond angle is less than the perfect 109.5 degrees seen in a tetrahedral molecule due to this repulsion. This distortion is essential in understanding the reactivity of PCl₃ in chemical syntheses and its behavior in different solvents.
What is the difference between electron geometry and molecular geometry?
Electron geometry refers to the arrangement of all electron pairs (bonding and non-bonding) around the central atom, while molecular geometry considers only the atoms bonded to the central atom, excluding lone pairs.
How does PCl₃'s lone pair affect its reactivity?
The lone pair makes PCl₃ a Lewis base, capable of donating a pair of electrons in chemical reactions, thus affecting its reactivity and interaction with other molecules.
The comprehension of PCl₃’s electron geometry not only elucidates its molecular structure but also offers predictive power for its chemical behavior and reactivity, making it indispensable knowledge for anyone delving into advanced chemical studies.