Have you ever wondered what happens when you mix different substances? Whether you're a science enthusiast, a student, or just curious, understanding the intricacies of chemical reactions can be fascinating and incredibly useful. The concept of reactants and products is fundamental to chemistry, impacting everything from cooking to industrial processes. This guide will walk you through everything you need to know to master the basics and tackle more complex concepts with confidence.
Understanding Reactants and Products: The Foundation of Chemical Reactions
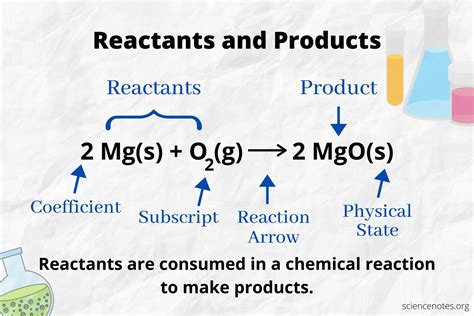
At the heart of every chemical reaction are two key components: reactants and products. Reactants are the starting materials involved in a reaction, and products are what you end up with after the reaction has taken place.
Let’s dive into a practical example to bring this concept to life. Imagine baking a cake. The ingredients you mix together—flour, sugar, eggs, and baking powder—are your reactants. Once mixed and baked, the end product is the delicious cake itself.
Problem-Solution Opening Addressing User Needs
Despite knowing the basic definitions, many struggle to grasp the nuanced roles of reactants and products in different types of reactions, which can hinder their understanding of chemistry as a whole. This guide will address these challenges by providing step-by-step guidance, real-world examples, and practical solutions to make complex concepts straightforward. You’ll discover how to predict what products will form, how to recognize different types of reactions, and how to avoid common mistakes. By the end of this guide, you’ll have a solid foundation in understanding chemical reactions and feel equipped to delve into more advanced topics.
Quick Reference
Quick Reference
- Immediate action item: When observing a chemical reaction, identify the reactants and products. This foundational skill will help you understand and predict outcomes.
- Essential tip: Use a balanced equation to represent a reaction. This helps ensure that the law of conservation of mass is maintained and that you accurately represent the transformation of reactants into products.
- Common mistake to avoid: Confusing reactants with products. Always remember that reactants are what you start with, and products are what you get at the end of the reaction.
Types of Chemical Reactions
Before we delve deeper, let’s explore the different types of chemical reactions. Knowing the type of reaction can help you predict the products and understand the reactants’ behavior.
Here are four primary types of chemical reactions:
Synthesis Reactions
In synthesis reactions, two or more simple substances combine to form a more complex product. A classic example is the reaction between sodium (Na) and chlorine (Cl) to form sodium chloride (NaCl).
Example: 2Na + Cl2 → 2NaCl
Decomposition Reactions
Decomposition reactions involve a single compound breaking down into two or more simpler substances. A well-known example is the decomposition of potassium chlorate (KClO3) into potassium chloride (KCl) and oxygen gas (O2) when heated.
Example: 2KClO3 → 2KCl + 3O2
Single Replacement Reactions
In single replacement reactions, an element reacts with a compound, and an element in the compound is replaced by the reacting element. An example is zinc (Zn) reacting with hydrochloric acid (HCl) to produce zinc chloride (ZnCl2) and hydrogen gas (H2).
Example: Zn + 2HCl → ZnCl2 + H2
Double Replacement Reactions
Double replacement reactions occur when parts of two compounds are exchanged, making two new compounds. An example is the reaction between silver nitrate (AgNO3) and sodium chloride (NaCl) to form silver chloride (AgCl) and sodium nitrate (NaNO3).
Example: AgNO3 + NaCl → AgCl + NaNO3
How to Balance Chemical Equations
Balancing chemical equations is a critical skill in chemistry, ensuring the conservation of mass and providing a true representation of the reaction.
Here’s a step-by-step process:
Step 1: Write the Unbalanced Equation
Begin by writing out the chemical equation without balancing it. For example, for the combustion of propane (C3H8), the unbalanced equation is:
C3H8 + O2 → CO2 + H2OStep 2: Count the Number of Atoms
Count the number of each type of atom on both sides of the equation.
For propane combustion:
| Type of Atom | Reactant Side | Product Side |
|---|---|---|
| Carbon © | 3 | 2 |
| Hydrogen (H) | 8 | 2 |
| Oxygen (O) | 2 | 3 |
Step 3: Balance Carbon and Hydrogen
Since there are more carbons and hydrogens on the reactant side, you need to adjust the coefficients accordingly.
C3H8 + O2 → 3CO2 + 4H2O
Step 4: Balance Oxygen
The equation now has 9 oxygen atoms on the product side (3 from CO2 * 3 + 4 from H2O * 1). To balance, you need 5 O2 molecules on the reactant side:
C3H8 + 5O2 → 3CO2 + 4H2OHow to Identify the Type of Reaction
Identifying the type of chemical reaction is crucial for predicting the products and understanding the reactants’ behavior.
Follow these steps:
Step 1: Observe the Reactants and Products
Identify whether the reactants are combining or breaking apart, or if there’s a replacement happening.
Step 2: Use Clues from Chemical Formulas
Certain formulas give hints about the type of reaction:
- Synthesis: Two or more reactants form one product.
- Decomposition: One reactant breaks down into two or more products.
- Single Replacement: An element replaces another element in a compound.
- Double Replacement: Two compounds exchange components.
Step 3: Consider Energy Changes
Exothermic reactions release energy (like heat), and endothermic reactions absorb energy. While not always easy to determine without specific data, it can provide clues about the type of reaction.
Common Mistakes and How to Avoid Them
Avoiding common pitfalls is crucial for mastering chemical reactions. Here are some mistakes to watch out for:
Mistake 1: Overlooking the Law of Conservation of Mass
Ensure that your balanced equation reflects the law of conservation of mass, meaning the number of each type of atom on the reactant side equals the number on the product side.