Understanding the intricacies of strong acids and bases is essential for both scientific exploration and practical applications in everyday life. This guide aims to provide you with the actionable insights needed to navigate this complex topic with confidence and ease. Whether you are a student preparing for exams, a professional in a lab setting, or simply someone interested in the chemistry of acids and bases, this comprehensive guide will serve as your go-to resource.
Why Knowing Strong Acids and Bases is Important
Strong acids and bases are crucial in various scientific domains. They are often involved in chemical reactions that drive industries such as pharmaceuticals, agriculture, and environmental science. In practical terms, understanding these substances can also help in making informed decisions regarding the safety of household chemicals, environmental conservation, and even culinary practices.
Problem-Solution Opening Addressing User Needs
Many individuals find the concept of strong acids and bases daunting due to their complex chemistry and the potential risks they pose. This guide is designed to demystify these substances by breaking down their characteristics, behaviors, and uses into understandable and actionable segments. From avoiding common pitfalls to leveraging these chemicals in beneficial ways, this guide offers clear, step-by-step instructions to address your specific needs, ensuring you gain confidence and competence in handling these powerful compounds.
Quick Reference Guide
Quick Reference
- Immediate action item: If you spill a strong acid or base, immediately evacuate the area and call for professional help.
- Essential tip: Always use personal protective equipment (PPE) when handling strong acids and bases, including gloves, goggles, and lab coats.
- Common mistake to avoid: Assuming that “dilute” versions of strong acids and bases are safe—even small quantities can still cause significant harm.
Understanding Strong Acids: Detailed How-To Sections
To grasp the concept of strong acids, let’s dive into their characteristics, properties, and safe handling practices.
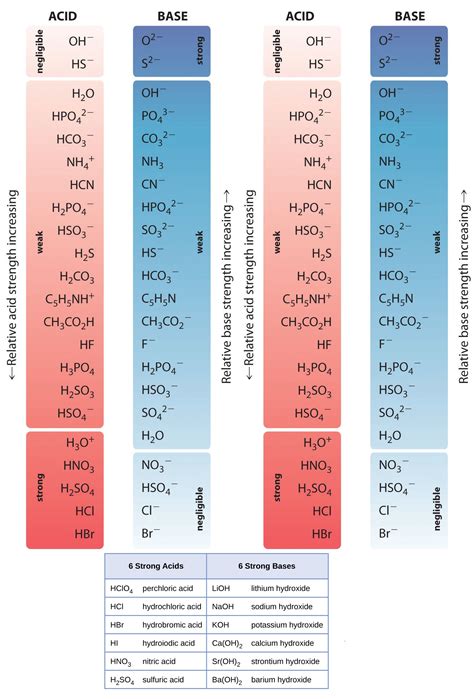
Strong acids are compounds that completely dissociate in water, releasing a high number of hydrogen ions (H+). This complete dissociation makes them potent and reactive. Examples include hydrochloric acid (HCl), sulfuric acid (H2SO4), and nitric acid (HNO3).
Characteristics of Strong Acids
Here’s a closer look at what makes strong acids so powerful:
- Complete Dissociation: Strong acids ionize completely when they dissolve in water. For instance, HCl in water dissociates into H+ and Cl- ions completely.
- pH Levels: They generally have a pH below 3 due to their high concentration of H+ ions.
- Reactivity: Strong acids are highly reactive, leading to vigorous reactions with metals, carbonates, and even some organic compounds.
Properties and Uses
The properties of strong acids make them invaluable in various applications:
- Industrial Uses: Strong acids are used in large-scale industrial processes such as metal cleaning, pickling of steel, and in the production of fertilizers.
- Laboratory Applications: They are crucial in the laboratory for pH adjustment, neutralizing bases, and as reagents in various chemical reactions.
Safe Handling Practices
Safety is paramount when dealing with strong acids due to their corrosive and harmful nature.
- Personal Protective Equipment (PPE): Always wear gloves, goggles, and lab coats when handling acids to protect your skin and eyes.
- Proper Storage: Store acids in clearly labeled containers, away from incompatible substances like bases and organic materials to avoid dangerous reactions.
- Spill Management: Have spill kits on hand that include neutralizing agents (like bases) and absorbent materials to manage accidental spills promptly.
Understanding Strong Bases: Detailed How-To Sections
Strong bases, also known as strong alkalis, are compounds that completely dissociate in water, releasing hydroxide ions (OH-). Their complete dissociation makes them similarly potent and reactive. Examples include sodium hydroxide (NaOH), potassium hydroxide (KOH), and calcium hydroxide (Ca(OH)2).
Characteristics of Strong Bases
Strong bases share some common characteristics that make them significant in both laboratory and industrial settings:
- Complete Dissociation: Strong bases fully dissociate into OH- and metal ions in water, similar to the complete dissociation of strong acids into H+ ions.
- pH Levels: They generally have a pH above 11 due to their high concentration of OH- ions.
- Reactivity: Strong bases are highly reactive, often leading to vigorous reactions with acids, metals, and some organic compounds.
Properties and Uses
The properties of strong bases make them valuable in a variety of applications:
- Industrial Uses: Strong bases are used in manufacturing, paper production, and in the production of cleaning products and detergents.
- Laboratory Applications: They are essential for pH adjustment, neutralizing acids, and as reagents in various chemical reactions.
Safe Handling Practices
Just like strong acids, the safe handling of strong bases is crucial:
- Personal Protective Equipment (PPE): Always wear gloves, goggles, and lab coats to protect against corrosive skin and eye damage.
- Proper Storage: Store bases in clearly labeled containers, away from acids and organic materials to prevent dangerous reactions.
- Spill Management: Have spill kits on hand that include neutralizing agents (such as acids) and absorbent materials to manage accidental spills efficiently.
Practical FAQ Section
Common user question about practical application
How can I neutralize a strong acid or base safely?
Neutralizing acids and bases safely involves a few key steps:
- Identify the substance: Determine whether you are dealing with a strong acid or base. This is crucial as the neutralizing agent will vary accordingly.
- Use appropriate neutralizing agents: For strong acids, use a strong base such as sodium hydroxide. Conversely, for strong bases, use a strong acid like hydrochloric acid. Always use in small amounts to avoid vigorous reactions.
- Dilute carefully: Slowly add the neutralizing agent to the acid or base while stirring. Never the other way around to prevent splashing and exothermic reactions.
- Dispose of properly: Once neutralized, dispose of the chemical waste according to local regulations. Never pour neutralized acids or bases down the drain.
- Personal Protection: Always wear appropriate PPE including gloves, goggles, and lab coats during this process to protect yourself from chemical splashes and reactions.
Conclusion
Understanding strong acids and bases is a critical skill that bridges both theoretical knowledge and practical application across various fields. This guide provided a clear, step-by-step exploration of these powerful chemicals, emphasizing safety and practical use. Armed with this knowledge, you can confidently navigate and utilize strong acids and bases in a safe, efficient, and informed manner.