When delving into molecular structures, the trigonal bipyramidal shape often emerges, posing both challenges and insights for chemists and students alike. This molecular geometry stands as a critical component in fields such as inorganic chemistry, materials science, and beyond. However, grasping the intricacies of trigonal bipyramidal bond angles is not always straightforward. This guide aims to demystify these complexities through actionable advice, real-world examples, and a conversational tone that’s accessible to both novice and seasoned chemists.
Understanding the Trigonal Bipyramidal Geometry
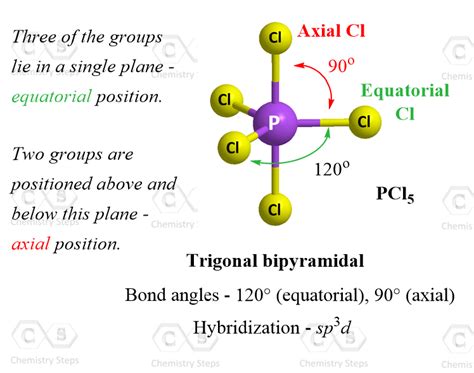
The trigonal bipyramidal geometry is characterized by a central atom bonded to five peripheral atoms arranged in a three-dimensional configuration. This shape may appear intimidating at first glance, but with a structured approach, understanding it becomes both manageable and fascinating. In a trigonal bipyramidal arrangement, the central atom is surrounded by three equatorial atoms positioned at 120° angles to each other and two axial atoms positioned directly above and below the central atom at an angle of 90° to the equatorial plane.
Problem-Solution Opening: Addressing Your Needs
If you’re navigating the complexities of trigonal bipyramidal bond angles, you’re likely looking for a reliable, step-by-step guide that makes molecular mysteries more transparent. This guide is tailored to answer your most pressing questions, offering actionable advice to help you understand the geometric and chemical implications of this molecular structure. Whether you’re dealing with VSEPR theory, molecular orbitals, or predicting the behavior of specific compounds, we’ve got you covered. By breaking down the theory and providing real-world examples, we aim to transform what could be a daunting task into a manageable and intellectually rewarding challenge.
Quick Reference
Quick Reference
- Immediate action item: Draw the trigonal bipyramidal structure and label the bond angles to see the spatial arrangement.
- Essential tip: Remember that the equatorial positions allow for a larger 120° bond angle compared to the 90° angles at the axial positions.
- Common mistake to avoid: Confusing the positions of equatorial and axial atoms. Make sure to place the two axial atoms directly above and below the central atom.
Detailed How-To Sections: Exploring Trigonal Bipyramidal Bond Angles
Let’s dive deeper into the trigonal bipyramidal bond angles, starting with an explanation of VSEPR theory and moving towards specific examples.
VSEPR Theory and Its Application
Valence Shell Electron Pair Repulsion (VSEPR) theory provides a straightforward method to predict the geometry of molecules. According to VSEPR theory, electron pairs around a central atom will arrange themselves to minimize repulsion. For a trigonal bipyramidal structure:
- There are five pairs of electrons around the central atom.
- These electron pairs spread out in space to minimize repulsion, leading to the trigonal bipyramidal arrangement.
- Equatorial positions offer the most space and thus accommodate the three pairs of electrons, leading to 120° angles between them.
- The axial positions, being more confined, host the other two pairs of electrons with a 90° angle to each other and the equatorial atoms.
Detailed Calculation and Examples
Let’s illustrate the trigonal bipyramidal bond angles with an example: Phosphorus pentachloride (PCl5).
PCl5 is a classic example of a molecule adopting a trigonal bipyramidal shape. Here’s how you can analyze its bond angles:
- Step 1: Identify the central atom. In PCl5, phosphorus (P) is the central atom.
- Step 2: Count the electron pairs around the central atom. PCl5 has five pairs of electrons.
- Step 3: Predict the geometry. Since there are five pairs, the molecule will adopt a trigonal bipyramidal geometry.
- Step 4: Determine the bond angles. Equatorial-equatorial pairs will be 120° apart, and axial-axial pairs will be 90° apart.
Real-World Examples
Understanding trigonal bipyramidal structures through examples helps solidify your grasp. Let’s consider two more examples:
- IF5: Iodine pentafluoride features iodine as the central atom surrounded by five fluorine atoms, forming a trigonal bipyramidal shape.
- IF7: If you're curious about how this geometry can extend, Iodine heptafluoride initially forms a trigonal bipyramidal structure with one axial position occupied by a lone pair, which significantly affects its shape and properties.
Practical FAQ
What are the bond angles in a trigonal bipyramidal molecule?
In a perfect trigonal bipyramidal geometry, the bond angles between the equatorial atoms are 120°. The angles between the axial atoms and their nearest equatorial neighbors are also 90°, while the angles between the axial and equatorial atoms are close to 90°.
How can I visually confirm the trigonal bipyramidal arrangement?
Visual confirmation can be achieved by drawing the structure or using molecular model kits. Begin by placing the central atom and then arrange the five peripheral atoms to form the trigonal bipyramidal shape. Label the bond angles accordingly to understand their spatial arrangement and relative measurements.
Why does the bond angle differ between equatorial and axial positions?
The difference in bond angles between equatorial and axial positions is due to the spatial arrangement dictated by VSEPR theory. Equatorial positions have three pairs of electrons providing the maximum space, leading to 120° angles. Axial positions are more confined with only two pairs, leading to 90° angles. This arrangement minimizes electron pair repulsion, stabilizing the molecule.
By mastering the trigonal bipyramidal geometry and bond angles, you’ll gain valuable insight into a wide range of chemical compounds and their properties. This detailed guide and practical examples should equip you with the knowledge to approach molecular structures with confidence and clarity.