Understanding the complexities of protein monomers is paramount in advancing biotechnological and biomedical research. At the core of all cellular functions lies the intricate network of proteins, which perform an array of tasks necessary for life. Protein monomers are the building blocks of these larger structures, forming complex networks and functions within organisms. This article delves into the nuances of protein monomers, providing expert insights, practical examples, and actionable recommendations to aid in the broader application of this knowledge in research and development.
Key Insights
- Protein monomers form the foundation of protein structure and function.
- Understanding the folding and interactions of monomers is critical for advanced drug development.
- Practical applications include developing targeted therapies for diseases associated with protein misfolding.
The Fundamental Nature of Protein Monomers
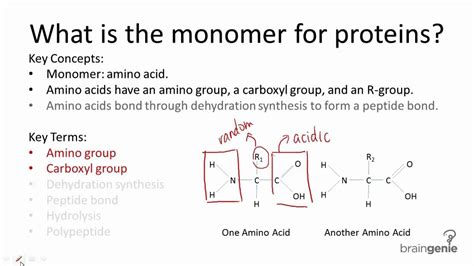
Proteins are polymers composed of amino acid monomers, each of which brings its own set of properties. The sequence in which these monomers are arranged determines the protein’s structure and function. The process by which monomers fold into functional three-dimensional structures is a fundamental area of study in biochemistry. Errors in this process can lead to a host of diseases, including neurodegenerative disorders like Alzheimer’s and Parkinson’s disease.The folding process is driven by various intramolecular forces including hydrogen bonds, disulfide bridges, hydrophobic interactions, and van der Waals forces. The understanding of these interactions is essential for researchers aiming to manipulate protein folding, either for therapeutic purposes or to enhance protein stability for industrial applications.
Applications in Drug Development
The significance of protein monomers in drug development cannot be overstated. By understanding the interactions and folding patterns of monomers, scientists can design small molecules that specifically target aberrant protein structures, potentially correcting their misfolding and restoring function. This approach, known as molecular chaperone therapy, has shown promise in treating diseases where protein aggregation plays a crucial role.One example of this is the development of drugs for cystic fibrosis. This genetic disorder stems from mutations in the CFTR protein, which misfolds and fails to function correctly. Pharmaceutical companies are now researching small molecules that can bind to and stabilize the mutated CFTR, enabling it to function properly and alleviating symptoms.
How can understanding protein monomers lead to new therapies?
A deeper understanding of protein monomer interactions allows scientists to develop targeted therapies that correct protein misfolding and aggregation. This approach is crucial in treating diseases like Alzheimer's, Parkinson's, and cystic fibrosis.
What are the challenges in studying protein monomers?
One of the primary challenges lies in the complexity of protein folding and the transient nature of some interactions. Advanced techniques like cryo-electron microscopy and nuclear magnetic resonance (NMR) spectroscopy are used to study these processes at an atomic level.
The study of protein monomers is a rapidly evolving field that bridges fundamental biochemistry with practical applications in medicine and biotechnology. As research progresses, the ability to manipulate protein structure will continue to open new avenues for innovative therapies and industrial applications. This knowledge not only promises to unlock the secrets of cellular biology but also drives forward the frontier of human health and technology.