Understanding chemiosmosis is key to unlocking the mysteries of cellular energy production, a fundamental process for all life forms. The principle of chemiosmosis lies at the heart of how cells generate the energy they need to perform vital functions. This guide will walk you through the step-by-step process, offering actionable advice and practical solutions to help you fully grasp this complex concept. Let’s dive in and demystify the science behind cellular energy production.
Why Chemiosmosis Matters
Chemiosmosis is a foundational biological process that allows cells to harness energy from electrochemical gradients to synthesize ATP, the energy currency of the cell. Without chemiosmosis, cells wouldn't be able to perform essential functions like growth, repair, and reproduction. This guide will provide a clear understanding of how chemiosmosis works, its significance, and practical examples to help you integrate this knowledge into both academic study and practical applications in biotechnological advancements.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: To understand the basic principle of chemiosmosis, start with the proton motive force, the driving force behind ATP synthesis.
- Essential tip with step-by-step guidance: Follow the electron transport chain, and note how it creates the proton gradient that fuels ATP synthesis.
- Common mistake to avoid with solution: Confusing chemiosmosis with substrate-level phosphorylation. Remember, chemiosmosis relies on a proton gradient, while substrate-level phosphorylation occurs directly through substrate reactions.
Step-by-Step Guide to Understanding Chemiosmosis
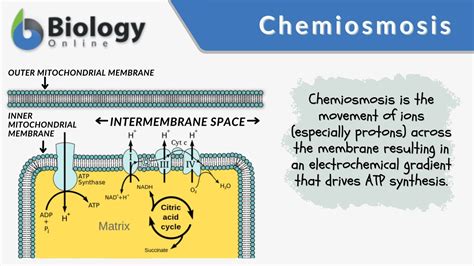
Chemiosmosis refers to the process by which ATP is synthesized thanks to the flow of protons across a semipermeable membrane. Here, we'll break down the process into digestible steps.
Step 1: The Electron Transport Chain
Begin by understanding the electron transport chain (ETC), a series of protein complexes in the inner mitochondrial membrane in eukaryotic cells or the plasma membrane in prokaryotic cells. The ETC plays a crucial role in generating the proton gradient:
- Electrons are transferred from electron carriers like NADH and FADH2 to oxygen via a series of redox reactions.
- These reactions pump protons (H+) from the mitochondrial matrix into the intermembrane space, creating a proton gradient.
Step 2: Formation of the Proton Gradient
The proton gradient formed by the ETC is a key component of chemiosmosis:
- As protons are pumped into the intermembrane space, the matrix becomes more negative relative to the intermembrane space.
- This creates an electrochemical gradient, or proton motive force (PMF).
Step 3: ATP Synthesis via ATP Synthase
The PMF drives the synthesis of ATP in a process facilitated by ATP synthase, a complex enzyme:
- Protons flow back into the mitochondrial matrix through ATP synthase, driven by the electrochemical gradient.
- As protons pass through ATP synthase, it rotates and catalyzes the conversion of ADP and inorganic phosphate (Pi) into ATP.
Step 4: The Role of Chemiosmosis in Different Organisms
Chemiosmosis occurs in various forms across different organisms:
- In plants, chemiosmosis occurs during photosynthesis in the thylakoid membrane, where light energy generates the proton gradient.
- In bacteria, it occurs in the plasma membrane, utilizing substrate-level processes to create the gradient.
Detailed How-To Sections
How to Identify the Key Components of Chemiosmosis
Recognizing the main components of chemiosmosis is crucial for understanding the whole process:
- Start by identifying the electron transport chain and ATP synthase. These are the primary players in the chemiosmotic process.
- Pay attention to the proton gradient and the semipermeable membrane, which are the physical drivers of the process.
How to Visualize the Chemiosmotic Process
Visualization aids in comprehension. Here’s how you can better visualize chemiosmosis:
- Use diagrams that show the proton movement and ATP synthesis. Visualization tools can simplify complex processes.
- Consider interactive models available online, which allow you to see the dynamic nature of proton flow and ATP production.
How to Apply Chemiosmosis Principles in Practical Research
Understanding chemiosmosis is not just academic; it has practical applications:
- Study the impact of inhibitors like oligomycin, which blocks ATP synthase and disrupts ATP production.
- Experiment with uncouplers such as 2,4-dinitrophenol, which dissipate the proton gradient and reduce ATP production efficiency.
Practical FAQ
What is the difference between chemiosmosis and oxidative phosphorylation?
Chemiosmosis specifically refers to the generation of ATP via the proton gradient, while oxidative phosphorylation encompasses the entire process of ATP production through the electron transport chain and chemiosmosis. In simple terms, oxidative phosphorylation includes the electron transport chain and ATP synthase, while chemiosmosis focuses on the gradient and ATP synthesis aspect.
How does chemiosmosis relate to photosynthesis in plants?
In photosynthesis, chemiosmosis occurs in the thylakoid membrane of chloroplasts. Light energy drives the electron transport chain, creating a proton gradient across the thylakoid membrane. This gradient then drives ATP synthesis via ATP synthase, which is crucial for the plant’s energy needs.
Best Practices to Master Chemiosmosis
Here are several best practices to solidify your understanding of chemiosmosis:
- Regularly review the electron transport chain and its role in proton gradient formation.
- Practice diagramming the proton gradient and ATP synthesis process to reinforce your visual understanding.
- Engage with real-world examples and experimental data to see chemiosmosis in action.
By following these detailed steps, essential tips, and best practices, you’ll develop a robust understanding of chemiosmosis and its pivotal role in cellular energy production.