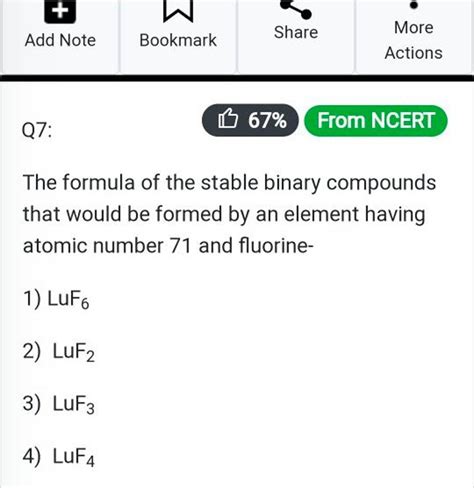
Unveiling the Secrets of Stable Binary Compounds
The world of chemistry is vast and full of intricate details that shape our understanding of the physical universe. Among the myriad of compounds studied, binary compounds hold a special place due to their straightforward composition, typically consisting of two elements. This article delves into the nature, formation, and stability of stable binary compounds, offering practical insights for professionals in the field.
Key Insights
- Binary compounds exhibit high stability due to strong ionic or covalent bonds, influencing their reactivity and applications.
- Understanding the electronic configuration is crucial for predicting the stability and type of bond formed between elements.
- Implementing computational chemistry methods can provide insights into the stability and properties of binary compounds, offering practical applications in material science.
Binary compounds are formed when two elements combine in fixed proportions, either through ionic or covalent bonding. The stability of these compounds largely depends on the nature of the bond formed. In ionic compounds, the stability is a result of the electrostatic forces between oppositely charged ions, exemplified by compounds like sodium chloride (NaCl). On the other hand, covalent binary compounds, such as water (H2O), are characterized by the sharing of electron pairs between atoms, leading to robust molecular structures.
The formation of stable binary compounds involves a delicate balance between attractive and repulsive forces at play. Ionic stability, for instance, is profoundly influenced by the charge density of the ions involved. Higher charge density typically leads to stronger electrostatic attraction and greater stability. This principle is evident in the high melting and boiling points of salts compared to molecular compounds.
For example, calcium oxide (CaO) demonstrates exceptional stability due to the strong ionic bonds formed between calcium and oxygen. The calcium ion (Ca²⁺) and oxide ion (O²⁻) have high charge densities, resulting in a highly stable compound with a melting point of 2572°C. This stability makes CaO a crucial material in industrial processes, including cement production.
In contrast, covalent binary compounds, such as carbon dioxide (CO₂), exhibit stability through the strong covalent bonds between carbon and oxygen atoms. These bonds are formed by the sharing of electrons, creating a linear molecule with symmetrical electron distribution. This molecular symmetry contributes to its stability and low reactivity at standard conditions.
In practical applications, stable binary compounds are invaluable across various fields, including material science, pharmaceuticals, and environmental science. The stability of these compounds ensures their reliability in applications ranging from constructing infrastructure to developing new medical treatments.
To delve deeper into the study of stable binary compounds, professionals can benefit from computational chemistry. Techniques such as density functional theory (DFT) can predict the stability and electronic properties of binary compounds by simulating their molecular structures and interactions. For example, computational studies of titanium dioxide (TiO₂) have revealed insights into its photocatalytic properties, paving the way for its use in environmental remediation and solar energy applications.
How do ionic and covalent bonds differ in their contribution to the stability of binary compounds?
Ionic bonds in binary compounds like NaCl are characterized by strong electrostatic attraction between oppositely charged ions, leading to high stability and typically high melting and boiling points. Covalent bonds, as seen in H2O, involve the sharing of electron pairs between atoms, resulting in stable molecular structures with different physical properties compared to ionic compounds.
What role does electronic configuration play in the stability of binary compounds?
Electronic configuration is crucial in determining the stability of binary compounds. Elements tend to form compounds that achieve a stable, full outer electron shell, often resembling the nearest noble gas configuration. This drive to achieve stability influences the type of bond formed (ionic or covalent) and the overall stability of the resulting compound.
Understanding the nature and stability of stable binary compounds equips professionals with the knowledge to leverage these materials in innovative and effective ways, underscoring their importance in both theoretical and applied chemistry.