Understanding and Calculating KCL Molar Mass: A Comprehensive Guide for Science Students
Welcome to your ultimate guide on determining KCL (potassium chloride) molar mass. Whether you're in high school or university, mastering the calculation of molar mass is crucial for your science education. This guide will walk you through the fundamentals, providing step-by-step instructions and practical examples to make the concept clear and accessible.
Knowing how to calculate molar mass will not only help you in exams but also equip you with a vital skill that is frequently used in chemical research and industrial applications. Let's dive right into it!
Why Understanding KCL Molar Mass Matters
Calculating the molar mass of compounds like potassium chloride (KCL) is fundamental in chemistry for determining how much substance is present in a given amount. This calculation is essential for various applications such as balancing chemical equations, determining reaction yields, and understanding stoichiometry. A precise understanding of these concepts will ensure your success in both academic and professional chemistry fields.
The molar mass of a compound is the mass in grams of one mole of that substance. For KCL, this value is determined by adding the atomic masses of potassium (K) and chlorine (Cl). Despite its simplicity, errors in calculation can lead to significant mistakes in chemical experiments.
Quick Reference: Key Points in Calculating KCL Molar Mass
Quick Reference
- Immediate action item: Look up the atomic masses of potassium and chlorine on the periodic table.
- Essential tip: Use a periodic table that lists atomic masses to ensure accuracy.
- Common mistake to avoid: Confusing atomic mass with molecular mass or mass percentage.
Step-by-Step Guide to Calculate KCL Molar Mass
Let's break down the process of calculating KCL molar mass step by step.
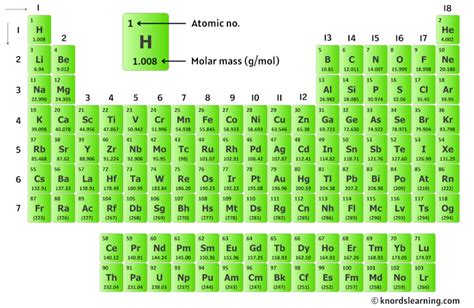
Step 1: Identify the atomic masses of potassium (K) and chlorine (Cl). These values can be found on a reliable periodic table. As of now, the atomic mass of potassium is approximately 39.10 g/mol, and that of chlorine is about 35.45 g/mol.
Step 2: Sum the atomic masses. To find the molar mass of KCL, add the atomic mass of potassium to the atomic mass of chlorine:
39.10 g/mol (K) + 35.45 g/mol (Cl) = 74.55 g/mol
Step 3: Verify your calculation. Double-check the addition to avoid any simple arithmetic mistakes. This precision is critical when dealing with chemicals.
To make this process even clearer, let’s dive deeper into each step with detailed examples and practical scenarios.
Detailed How-To: Step-by-Step Calculation
Identifying Atomic Masses
First, it’s important to know where to find the atomic masses. The periodic table is your best friend for this. Atomic masses are usually listed below the element’s symbol.
For instance, in a standard periodic table, potassium (K) might be located in group 1 and period 4, with its atomic mass listed as 39.10. Similarly, chlorine (Cl), found in group 17 and period 3, lists its atomic mass as 35.45.
Here’s a snapshot of how you might locate these on a periodic table:
| Element | Symbol | Atomic Mass |
|---|---|---|
| Potassium | K | 39.10 |
| Chlorine | Cl | 35.45 |
It's good practice to use a reputable source for these values to ensure precision.
Adding Atomic Masses
Once you’ve identified the atomic masses, the next step is to sum them to get the molar mass of KCL. This step involves basic arithmetic:
- For potassium, the atomic mass is 39.10 g/mol.
- For chlorine, the atomic mass is 35.45 g/mol.
Add these together:
39.10 g/mol (K) + 35.45 g/mol (Cl) = 74.55 g/mol
This is the molar mass of KCL.
Verifying Calculations
Calculation accuracy is crucial in chemistry. To verify your result, consider these tips:
- Check each atomic mass value for accuracy.
- Re-do the addition to confirm there are no errors.
- Ask a peer or teacher to double-check your work.
Even small errors in atomic mass values can lead to significant discrepancies in your molar mass calculations.
Practical FAQ: Addressing Common Questions
What if I need the molar mass for other KCL variations?
If you encounter different variations of KCL compounds, such as KClO3 (potassium chlorate) or KClO4 (potassium perchlorate), you’ll need to add the atomic masses of each constituent element accordingly. For KClO3, you would add the molar mass of potassium (K), chlorine (Cl), and three oxygens (O).
<ul>
<li>Atomic mass of K = 39.10 g/mol</li>
<li>Atomic mass of Cl = 35.45 g/mol</li>
<li>Atomic mass of O (multiplied by 3) = 16.00 g/mol × 3 = 48.00 g/mol</li>
</ul>
<p>So, for KClO3:</p>
<p>39.10 g/mol (K) + 35.45 g/mol (Cl) + 48.00 g/mol (O × 3) = 122.55 g/mol</p>
<p>Follow this method for other compounds by identifying and adding the atomic masses of all constituent elements.</p>
</div>
To summarize, understanding and calculating KCL molar mass involves identifying the atomic masses of each element involved, summing them accurately, and verifying your results. This guide serves as your roadmap, providing practical examples and detailed steps to ensure comprehension and precision.
Keep practicing with different compounds to master this fundamental skill. With time, calculating molar mass will become second nature, aiding you in more complex chemistry problems and experiments.
Happy calculating!